Local complications are related to poor long-term outcome in patients undergoing curative gastrectomy for advanced gastric cancer
Article information
Abstract
Purpose
The present study was performed to investigate the effects of local complications (LC) on long-term survival and cancer recurrence in patients undergoing curative gastrectomy for gastric cancer.
Methods
We analyzed 2,627 patients after curative gastrectomy for gastric cancer between January 2001 and December 2006. Patients were classified into groups no complications (NC), LC, or systemic complications (SC).
Results
Among the 2,627 patients, 475 patients developed complications (LC group [n=374, 14.2%] and SC group [n=101, 3.9%]). The 5-year cancer-specific survival rate was significantly poorer in the LC group compared to the NC and SC groups (LC, 78.0%; NC, 85.4%; SC, 80.2%; P=0.007). The occurrence of LC was identified as a significant independent prognostic factor for overall and cancer-specific survival (hazard ratio [HR], 2.08; 95% confidence interval [CI], 1.46–2.97; P=0.001 and HR, 1.77; 95% CI, 1.12–2.81; P=0.015). The tumor recurrence rates were higher in the LC group than the in other two groups (LC, 23.5%; NC, 15.4%; SC, 15.8%; P<0.001). The occurrence of LC was an independent predictor of tumor recurrence in patients undergoing curative gastrectomy for gastric cancer (HR, 1.55; 95% CI, 1.11–2.17; P=0.011).
Conclusion
LC are associated with adverse long-term outcomes in patients after curative gastrectomy for advanced gastric cancer.
INTRODUCTION
Despite its decreasing incidence rate, gastric cancer remains a common malignancy worldwide. In particular, gastric cancer is the second most common cause of cancer death in Korea, although the number of patients diagnosed with early gastric cancer has increased due to the development of gastroscopy and mass screening tests [1]. Age, tumor size, tumor location, histological classification, depth of invasion, lymph node invasion, lymphatic and vascular invasion, and distant metastasis have been identified as prognostic factors that influence the treatment outcome of gastric cancer [1–3]. Of these, D2 lymphadenectomy is the sole treatment option depending on the skill of the attending physician [4].
Anastomotic leakage, which is one of the most serious complications after gastrointestinal surgery, is related to poor outcome despite improvements in surgical techniques and perioperative management of cancer surgery [5–7]. In esophageal and colon cancer surgery, several studies have reported that negative effects of complications, including anastomotic leakage, are associated with postoperative mortality, long-term outcome, and recurrence [5–7]. Some recent studies have investigated the relationship between postoperative complications and long-term oncological outcome in cases of stomach cancer surgery [8,9]. However, the impact of complications other than anastomotic leakage and intraabdominal infection were not assessed, and studies have been limited to cases in which only total gastrectomy was performed or in cases of advanced but not early gastric cancer [8,10].
This study was performed to investigate the impact of postoperative local and systemic complications (SC) on long-term survival and recurrence rates in patients following curative gastrectomy for gastric cancer.
METHODS
This was a retrospective multicenter observational cohort study of patients who underwent gastrectomy for gastric cancer between January 2001 and December 2006 at nine surgical centers: Soonchunhyang University Bucheon Hospital, Keimyung University Hospital, National Cancer Center, Dong-A University Hospital, Yonsei University Hospital, Chonbuk National University Hospital, Seoul National University Hospital, Hallym University Dongtan Sacred Heart Hospital, Samsung Medical Center, and Sungkyunkwan University. Consecutive patients who underwent gastrectomy for gastric cancer were prospectively registered over the study period. This study was granted Institutional Review Board approval (IRB No. SCHBC 2014-07-011) and informed consent was waived because of the retrospective observational design and the anonymous nature of the study.
Study population
The inclusion criteria required patients to be histologically diagnosed with gastric cancer and to be receiving surgical treatment during the study period. Patients diagnosed with synchronous primary malignancy, those undergoing non-curative surgery, and those receiving preoperative chemotherapy were excluded.
Definitions
Postoperative complications were defined by a combination of clinical findings based on symptoms and physical examination of patients, as well as the results of laboratory and radiological studies, including X-ray, ultrasonography, and computed tomography (CT). In this study, patients were divided into groups exhibiting no complications (NC), local complications (LC), or SC. LC included wound infection, fluid collection/abscess, intraabdominal bleeding, intraluminal bleeding, intestinal obstruction, ileus, stenosis, anastomotic leakage, pancreatitis, wound dehiscence, and cholecystitis. SC consisted of pulmonary, hepatic, cardiac, urinary tract infections, splenic infarction, transient ischemic attack, and delirium. The Clavien-Dindo classification was used to assess the severity of surgical complications, using the following definitions based on its contracted form: grade I: any complication not requiring medical or surgical treatment; grade II: a complication requiring pharmacological treatment but no active intervention; grade III: a complication requiring surgical, radiological, or endoscopic treatment; grade IV: complications that are potentially life-threatening and require intensive care. In this study, we proceeded to record the complications observed in our patients, distribute them according to the type of treatment required, and classify them on this scale during hospitalization after surgery. In case of wound repair, when the wound was approximated at bedside, it was classified as grade I, when repaired under local anesthesia in an operation theater as grade III. Severe complications were defined as those graded as >III [11]. Surgical methods were classified as subtotal gastrectomy (distal or proximal gastrectomy) and total gastrectomy (total or completion gastrectomy), carried out by standard radical gastrectomy with D1+ or more. No patients underwent laparoscopic gastrectomy in the present study. According to the Japanese Classification of Gastric Cancer second English edition [12], regional lymph node dissection was defined as follows: D1, dissection of perigastric lymph node (i.e., stations 1–6); D2, D1 dissection and additional removal of the nodes along the left gastric, common hepatic, splenic, and left hepatoduodenal arteries (i.e., stations 7–11). If gross residual tumor and microscopic tumor were absent in the primary tumor bed, the surgery was considered curative resection. T stage was defined according to the International Union Against Cancer TNM classification of malignant tumors seventh edition [13] as local tumor growth (T1, invasion of lamina propria or submucosa; T2, invasion of muscularis propria or subserosa; T3, penetration serosa without invasion of adjacent structures; and T4, invasion of adjacent structures). Definitions for N stages were as follows: N0, no regional lymph node metastasis; N1, metastasis in 1–6 regional lymph nodes: N2, metastasis in 7–15 regional lymph nodes; and N4, metastasis in more than 15 regional lymph nodes. The tumor biopsy specimens were histologically classified as differentiated (papillary adenocarcinoma, tubular adenocarcinoma of well or moderately differentiated type) or undifferentiated (poorly differentiated adenocarcinoma, signet-ring cell carcinoma, mucinous adenocarcinoma) carcinoma. Vascular invasion was defined as groups of tumor cells attached to the blood vessel wall and projecting into the lumen of blood vessels in the capsule or outside the tumor. Lymphatic invasion was defined as the presence of tumor emboli or tumor masses within lymphatic vessels. Body mass index (BMI) was defined as the patient’s weight in kilograms divided by height (in meters) squared. Patients were classified into two groups: normal weight (BMI <25 kg/m2) or overweight and obese (BMI >25 kg/m2) [14]. Recurrence patterns were classified as locoregional, peritoneal, and distant recurrence. Locoregional recurrence was defined as any cancer recurrence at the resection margin, operation bed within the region of the resection, or regional lymph node metastasis. Peritoneal recurrence was defined as peritoneal carcinomatosis or Krukenberg tumor. Distant recurrence was defined as metastasis to non-regional lymph node or systemic organs, such as the liver, lung, bone, brain, and ovaries.
Adjuvant chemotherapy and patient follow-up
The follow-up schedule and indications for adjuvant chemotherapy were not standardized among the various institutions. However, most patients with advanced gastric cancer at TNM stage II or higher received fluorouracil-based adjuvant treatment. No patients were given postoperative or perioperative radiotherapy. Follow-up observations were performed at 3- or 6-month intervals for the first 2 years, at 6-month intervals for the next 3 years, and then annually until the patient’s death. The follow-up program consisted of physical examination, laboratory blood tests, endoscopy, CT, and positron emission tomography-CT (PET-CT). Recurrence was diagnosed from imaging findings, including CT, magnetic resonance imaging, PET-CT, endoscopy, biopsies, or cytology.
Data collection
The following data were obtained from electronic medical records and the prospectively collected gastric cancer registry from each hospital: general characteristics of the patients, including demographic data, tumor site and size, type of surgical procedure, and extent of lymph node dissection. Overall survival was calculated from the time of surgery to death due to any cause, or to the last follow-up that ended without death. Cancer-specific survival was calculated as the date of a diagnosis of stomach cancer until date of death, and was censored if the primary or underlying cause of death was not stomach cancer. The primary outcome was the 5-year survival rate and the secondary outcome was the 5-year cancer recurrence rate.
Statistical analysis
All results and tables are presented as medians and interquartile ranges (IQRs) or as numbers (percentages) of patients because the majority of the data did not follow a normal distribution. The Mann-Whitney U test and Kruskal-Wallis one-way analysis of variance were used for comparison of continuous variables, and comparisons of categorical data were performed using the chi-squared or Fisher exact test where appropriate. Multivariate Cox regression analysis was used to determine independent predictors of the 5-year overall survival rate and the 5-year cancer recurrence rate in a forward stepwise manner. Covariates included univariate predictors with P<0.20 and those that we considered clinically relevant. Survival curves were constructed using Kaplan-Meier estimates and compared with the log-rank test. All tests were two-tailed and P<0.05 was considered statistically significant. Statistical tests were performed using SPSS software, version 18.0 for Windows (SPSS Inc., Chicago, IL, USA).
RESULTS
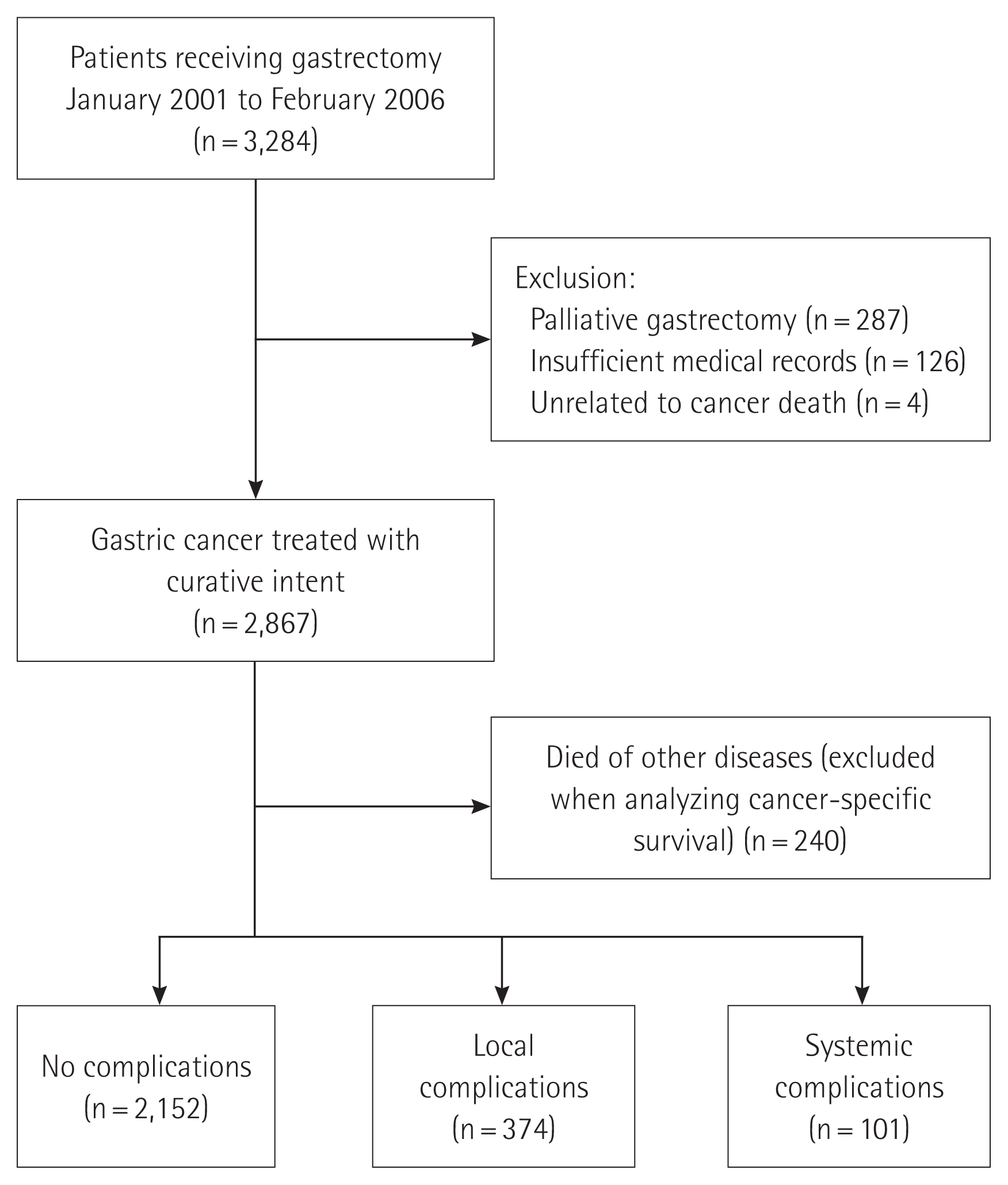
During the study period, a total of 3,284 patients who underwent gastrectomy for gastric cancer were entered into the registry. Of these, 417 patients were excluded according to the exclusion criteria: palliative gastrectomy (n=287), insufficient medical records (n=126), and reasons unrelated to cancer death (suicide and death due to traffic accidents, n=4). Ultimately, a total of 2,867 patients who underwent curative gastrectomy for gastric cancer were included in this study. Of the eligible patients, 240 (8%) who died of other diseases were excluded when analyzing cancer-specific survival. A total of 2,627 patients were categorized according to postoperative complication type into the NC group (n=2,152, 81.9%), the LC group (n=374, 14.2%), and the SC group (n=101, 3.8%) (Fig. 1).
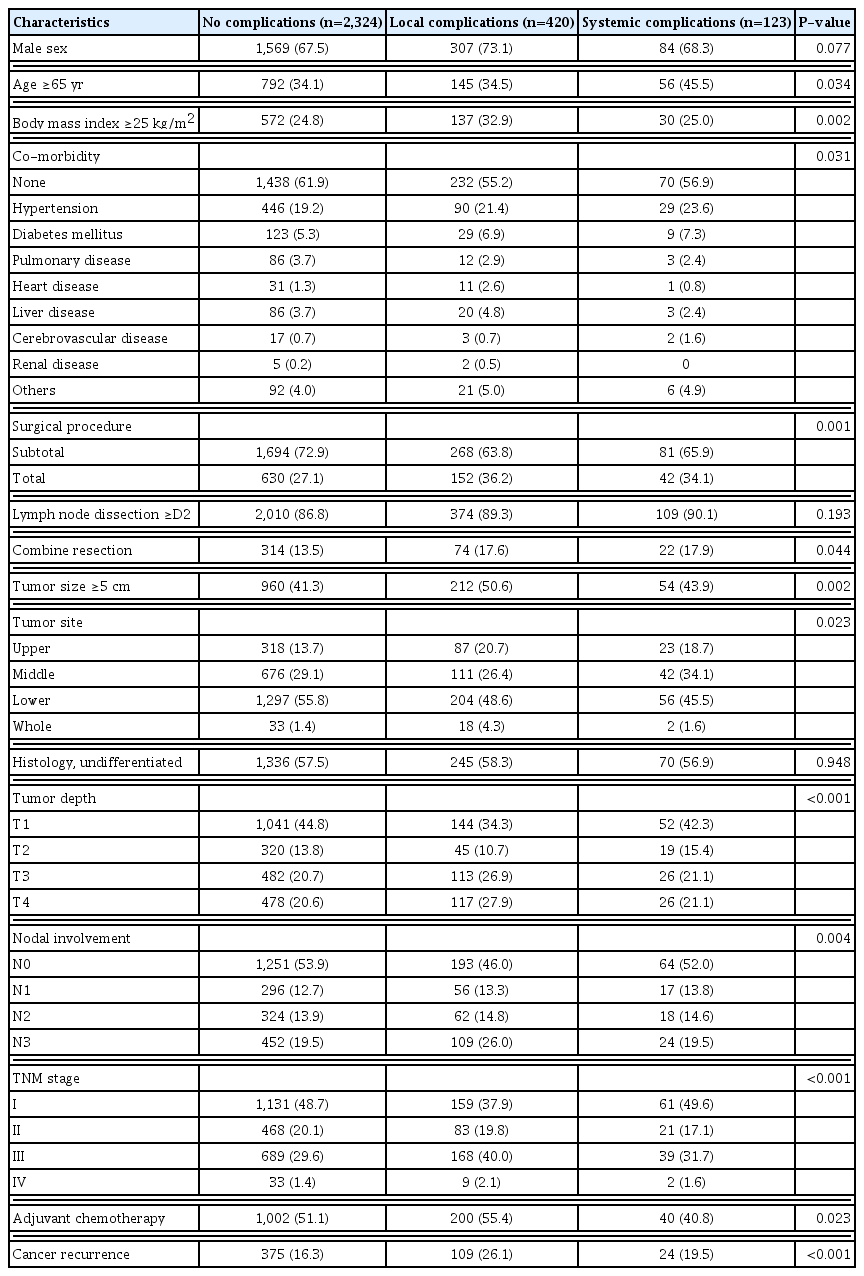
The clinicopathological characteristics of the 2,867 patients at the time of curative gastrectomy for gastric cancer are presented in Table 1. Overall, the patient population included 1,960 males (68.4%) and 907 females (31.6%), with a median age of 60 years (IQR, 50–67 years). The ratio of patients over age 65 was higher in the SC group. There were no significant differences between the three groups with respect to histological differentiation or lymph node dissection ≥D2. Total gastrectomy and combined resection were significantly more frequent in both the LC and the SC groups than in the NC group. BMI ≥25 kg/m2, tumor size ≥5 cm, advanced TNM stage, and recurrence of cancer were markedly higher in the LC group than in the other two groups.
Clinicopathological characteristics of 2,867 patients undergoing curative gastrectomy for gastric cancer
Of the 2,867 patients who underwent gastrectomy for gastric cancer, 543 patients (18.9%) developed complications. The numbers of Clavien-Dindo grade I, II, III, and IV complications were 155 (5.4%), 182 (6.3%), 132 (4.6%), and 74 (2.6%), respectively. Table 2 shows incidence of local and SC and grade ≥III according to the Clavien-Dindo classifications after curative gastrectomy. Among LC of grade III or over, wound problems, fluid collection/abscess, stenosis, and anastomotic leakage were relatively common. Most cases of anastomotic leakage required intervention (22/30 cases). Reoperation under general anesthesia was performed on 34 patients (19.5%) at grade III of LC group: intestinal obstruction (n=12), wound problems (n=8), anastomotic leakage (n=6), Fluid collection/abscess (n=3), intraabdominal bleeding (n=3), stasis/stenosis (n=2). As for SC, pulmonary and hepatic complications were common (2.1% and 0.5%, respectively).

Incidence of local and systemic complications and grade ≥III according to the Clavien-Dindo classification of 2,867 patients undergoing curative gastrectomy for gastric cancer
The median follow-up period of the surviving patients was 59.0 months (IQR, 48.0–73.0 months), and ranged from 1 to 106 months. The 5-year overall survival rate of the SC group was lower than those of the LC group and NC group (NC, 79.4%, LC, 70.7%; P<0.001, SC, 66.7%; P<0.001). The 5-year cancer-specific survival rates of NC, LC, and SC groups were 85.4%, 78.3% (P<0.001), and 80.2% (P=0.159), respectively, indicating that the occurrence of LC is related to poor survival (P=0.007) (Fig. 2). Fig. 3 shows a comparison of the 5-year cancer-specific survival rates among NC, LC, and SC groups according to TNM stage. The 5-year cancer-specific survival rates were 97.5% in the NC group, 96.7% (P=0.489) in the LC group, and 92.9% (P=0.062) in the SC group for patients with stage I disease; the differences among the three groups were not statistically significant (Fig. 3A). In stage II disease, the 5-year cancer-specific survival rates were 89.4%, 83.3%, and 84.6% in the NC, LC, and SC groups, respectively (P=0.011 and P=0.472), and those in stage III disease were 62.7%, 51.9%, and 59.0%, respectively (P=0.021 and P=0.244) (Fig. 3B and C). Thus, LC were related to significantly poor survival in patients receiving curative gastrectomy for advanced gastric cancer. In stage IV disease, the 5-year cancer-specific survival rates were 28.6% in the NC group and 0% in the LC group, and were not significantly different due to the small sample size (Fig. 3D).
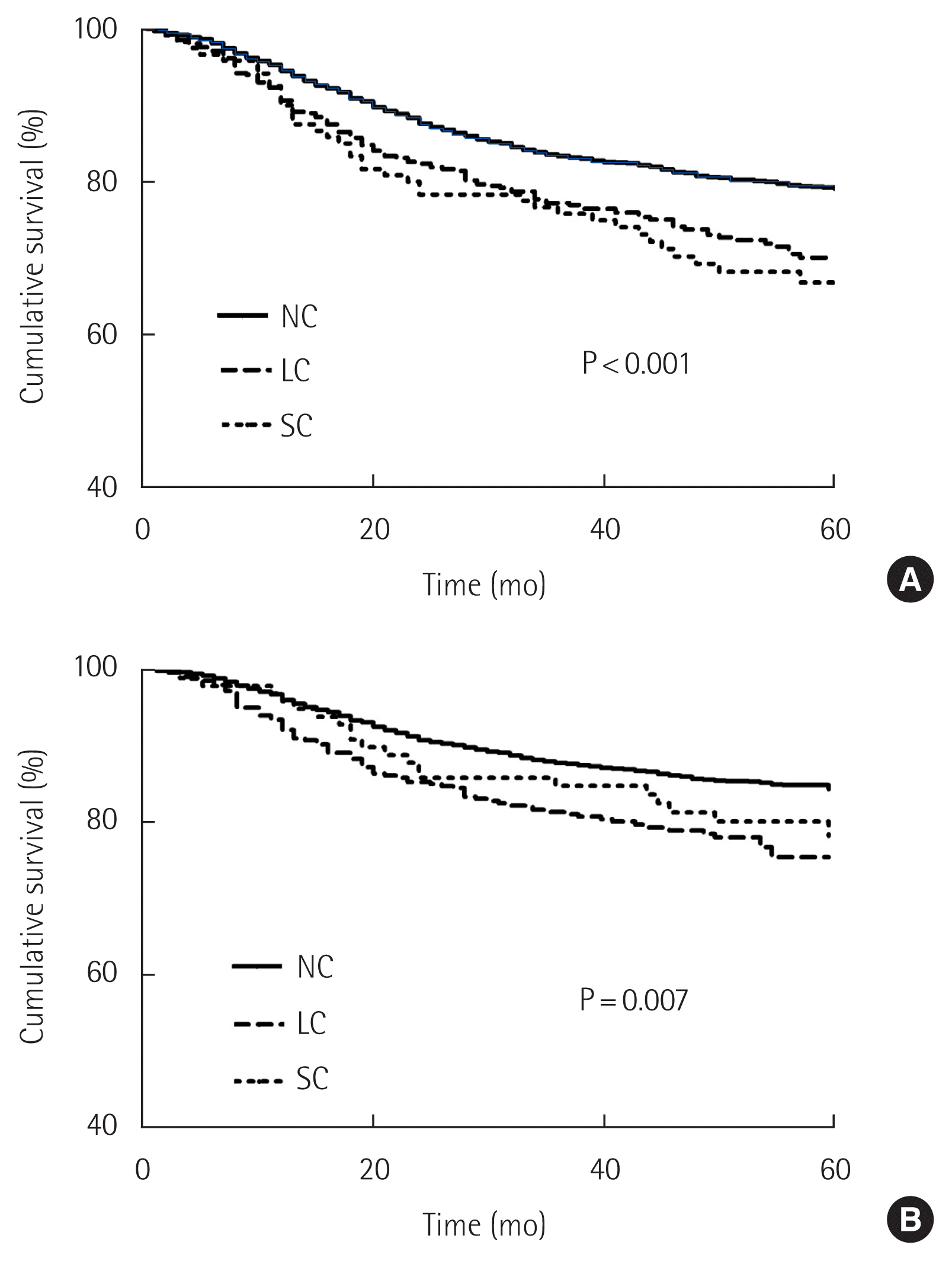
Comparison of 5-year overall survival and cancer-specific survival rates among groups with no complications (NC), local complications (LC), and systemic complications (SC). (A) Overall survival. (B) Cancer-specific survival.
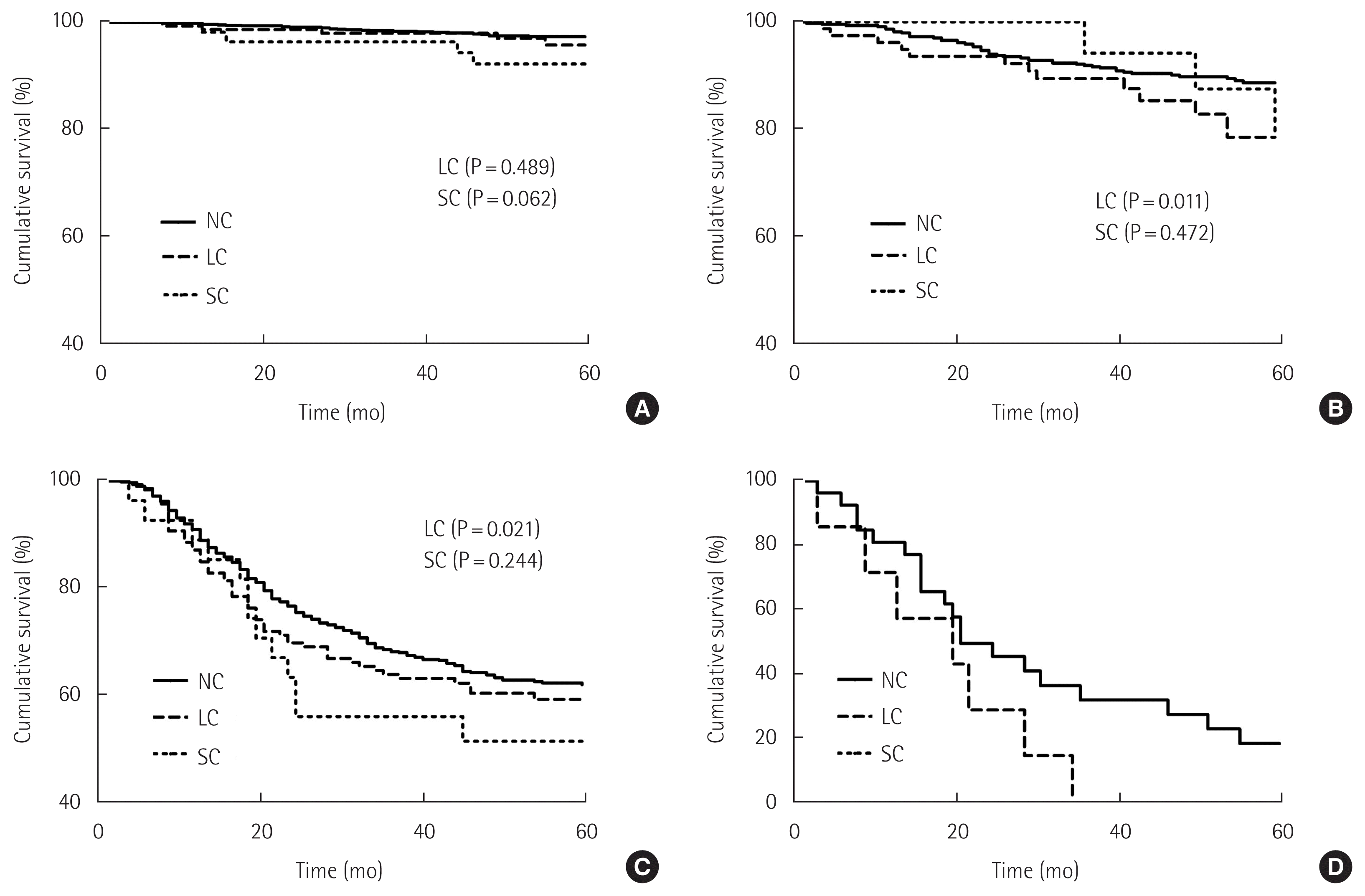
Comparison of the 5-year cancer-specific survival rates among groups with no complications (NC), local complications (LC) and systemic complications (SC) according to stage. (A) Stage I. (B) Stage II. (C) Stage III. (D) Stage IV.
In the analysis of patients in different severity of complications, increasing Clavien-Dindo scores from II to IV compared to no complication was significantly associated with a corresponding decrease in overall survival (80.4%, 71.4%, 66.5%, 29.5%, P<0.001) and cancer-specific survival (86.5%, 78.4%, 74.3%, 39.6% P< 0.001) (Fig. 4). The subgroup analysis showed that increasing Clavien-Dindo scores was associated with decreased overall survival (96.5%, 93.5%, 86.2%, 49.1%, P<0.001) and cancer-specific survival (98.5%, 96.2%, 90.8%, 74.7%, P<0.001) for stage II patients; decreased overall survival (82.7%, 76.3%, 70.3%, 39.5%, P<0.001) and cancer-specific survival (89.4%, 82.9%, 76.0%, 52.1%, P<0.001) for stage III patients; decreased overall survival (54.9%, 47.0%, 41.9%, 17.0%) and cancer-specific survival (62.6%, 55.9%, 49.2%, P<0.001) for stage IV patients.
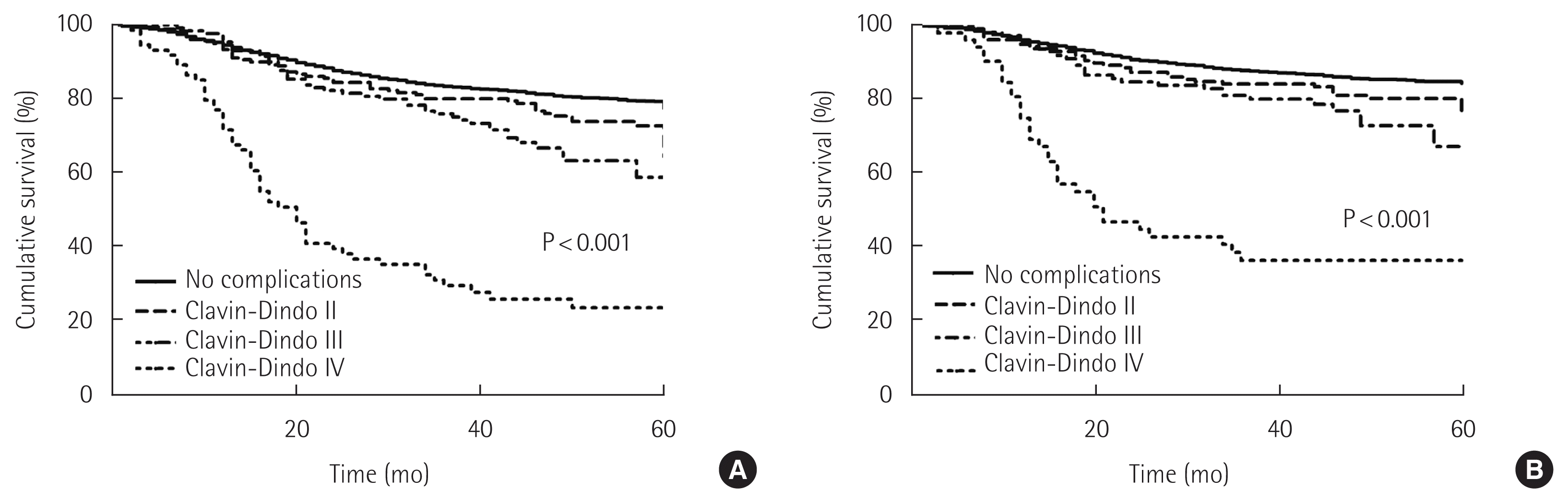
Comparison of survival outcomes based on the Clavien-Dindo classification. (A) Overall survival. (B) Cancer-specific survival.
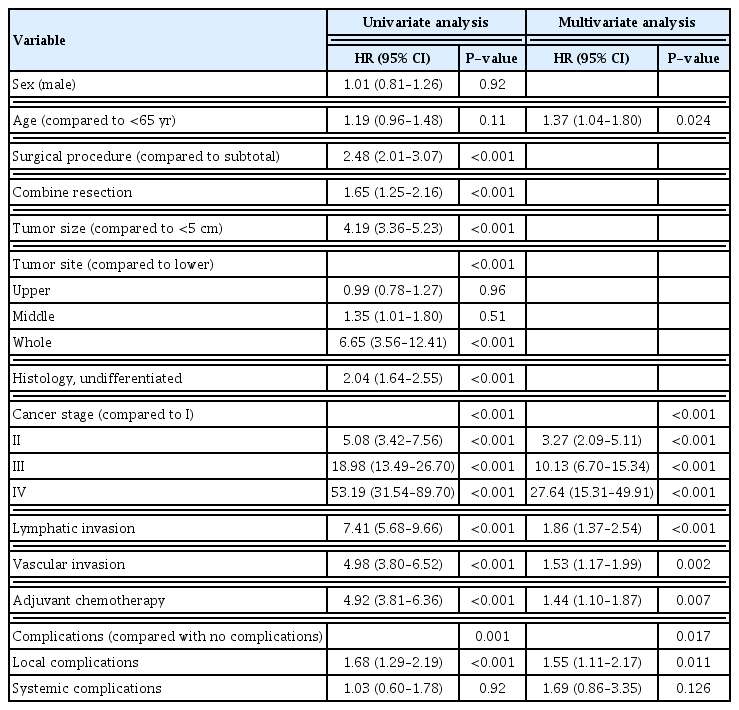
In the univariate analysis of the 5-year cancer-specific survival in 2,627 patients after curative gastrectomy for gastric cancer, age, total gastrectomy, combine resection, lymph node dissection ≥D2, tumor size, tumor in the whole stomach, histological differentiation, T category, N category, stage, lymphatic invasion, vascular invasion, adjuvant chemotherapy, and complication occurrence were identified as prognostic factors. Multivariate analysis of 5-year overall survival and cancer-specific survival rate after curative gastrectomy for gastric cancer are presented in Table 3. When multivariate analysis was performed for overall and cancer-specific survival rates, the common independent poor prognostic factors were patient age ≥65 years, tumor size ≥5 cm, total gastrectomy, lymph node dissection ≥D2, lymphatic invasion, vascular invasion, adjuvant chemotherapy, and advanced stage. In particular, the occurrence of LC was identified as a poor prognostic factor in the multivariate analysis of both overall and cancer-specific survival rates (overall survival: OR, 2.08; 95% CI, 1.46–2.97; P=0.001; cancer-specific survival: OR, 1.77; 95% CI, 1.12–2.81; P=0.015), but the presence of SC was not a significant factor (overall survival: OR, 1.21; 95% CI, 0.97–1.52; P=0.061; cancer-specific survival: OR, 1.26; 95% CI, 0.98–1.63; P=0.076).

Multivariate analyses of 5-year overall survival and cancer-specific survival rate after curative gastrectomy for gastric cancer
Recurrence rates were higher in the LC group (n=88, 23.5%) than in the NC and SC groups (n=332, 15.4% and n=16, 15.8%; P=0.017) (Fig. 5). Details of the initial recurrence site according to complication groups following curative gastrectomy are shown in Table 4. In the LC group, distant recurrence was the most frequent pattern, whereas peritoneal recurrence was the most frequently observed site of recurrence in the SC group. There were significant differences in patterns of recurrence between these three groups (P<0.001). Table 5 shows the results of univariate and multivariate analyses of factors associated with cancer recurrence. Multivariate analysis of the factors related to recurrence showed that age ≥65 years, advanced stage, lymphatic and vascular invasion, adjuvant chemotherapy, and LC were independent prognostic factors of recurrence.
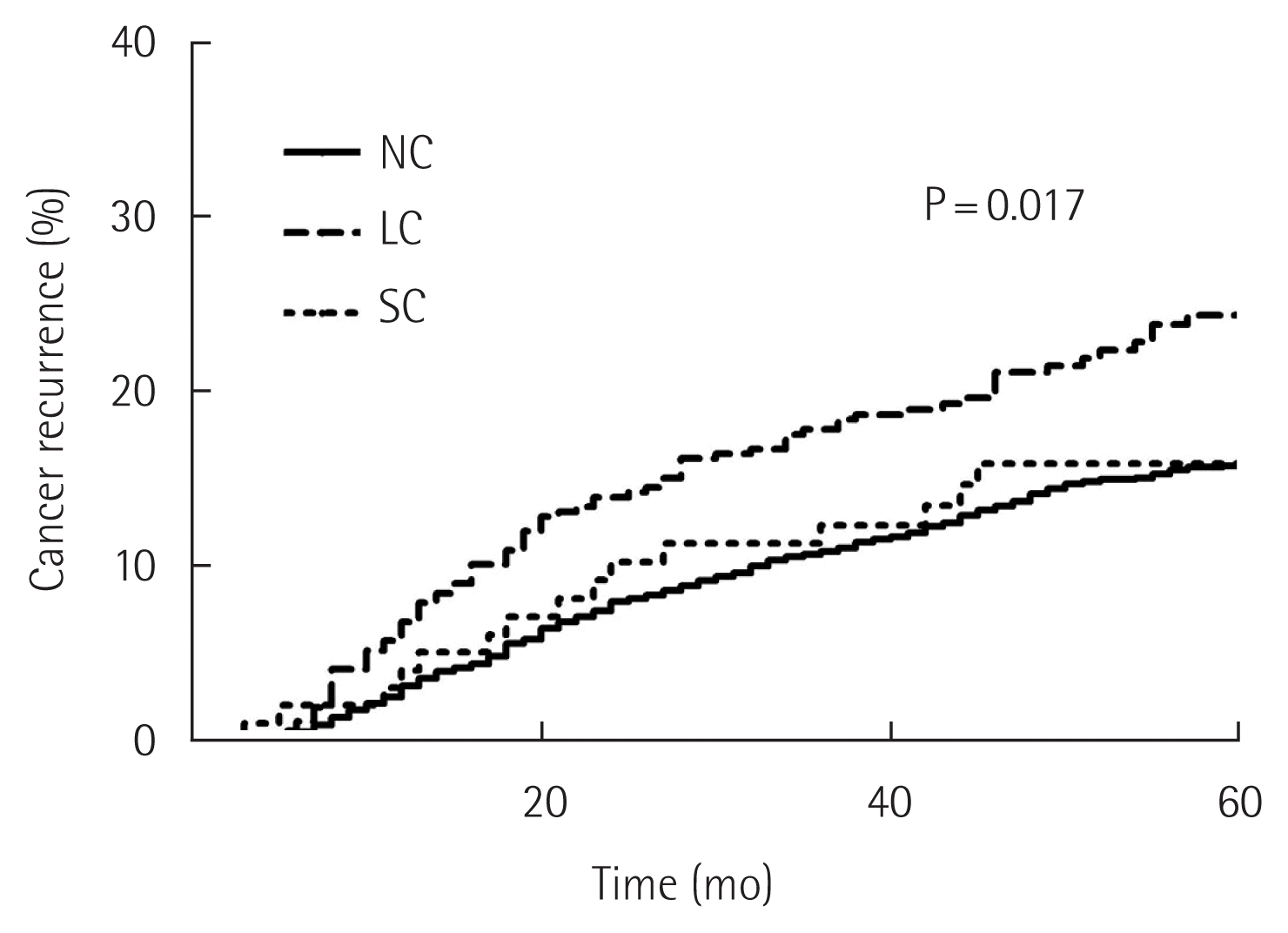
Comparison of 5-year cancer recurrence rates among groups with no complications (NC), local complications (LC) and systemic complications (SC) after curative gastrectomy for gastric cancer.

Initial recurrence site according to complications in 2,627 patients undergoing curative gastrectomy for gastric cancer
DISCUSSION
This is the largest multicenter study to examine the impact of postoperative complications on overall and cancer-specific survival rates in patients undergoing curative resection for gastric cancer. The results of the present study, along with previous reports in the literature, showed that LC following curative gastrectomy for gastric cancer are associated with high morbidity and mortality compared to other groups. In addition, the increase in the Clavien- Dindo classification proportionally deteriorate survival outcomes. Prognostic factors of the 5-year overall and cancer-specific survival in patients undergoing curative gastrectomy included LC as well as patient age, tumor size, total gastrectomy as the surgical procedure, more advanced stage of gastric cancer, vascular and lymphatic invasion, and adjuvant chemotherapy. Among the three groups, only the LC group was associated with higher recurrence rates, and the occurrence of LC was identified as one of the major independent prognostic factors of the 5-year cancer recurrence.
A number of recent studies have reported that the development of postoperative complications, especially intraabdominal sepsis, results in poorer long-term survival and increased risk of recurrence in patients undergoing resection of esophageal and colorectal cancer [6,15–18]. Sierzega et al. [8] showed for the first time in stomach cancer that anastomotic leakage has a negative impact on long-term survival after total gastrectomy for gastric cancer. However, their study included only total gastrectomy as the surgical procedure and also included the cases of non-curative gastrectomy, and did not investigate the impact of other postoperative complications, unlike our study. Tsujimoto et al. [9] reported a direct correlation between complications due to postoperative infection and unfavorable outcome, and anastomotic leakage was only a prognostic factor of cancer-specific survival although they limited infectious complications to pneumonia, enterocolitis, cholecystitis, anastomotic leakage, and intraabdominal abscess. In a recent study, Tokunaga et al. [19] reported poor long-term overall and relapse-free survival rates, and suggested that intraabdominal complications after curative gastrectomy increase mortality and exacerbate recurrence rates, even if infectious complications are restricted to anastomotic leakage, pancreas-related complications, and intraabdominal abscess. In the present study, however, we evaluated the relationships between all complications and 5-year overall and cancer-specific survival in patients after curative gastrectomy for gastric cancer of all stages. The results indicated that LC, but not SC, were significant predictors of poor outcome with regard to survival in patients who received curative gastrectomy for advanced but not early gastric cancer.
The incidence of postoperative complications is an important surrogate marker for measuring the quality of surgery. Dindo et al. [11] defined a complication as any deviation from the normal postoperative course and classified postoperative complications based on the type of therapy needed to correct the complication. The Clavien-Dindo classification system, which has proved to be a reliable tool for quality assessment in surgery, is adopted to overcome a difficult evaluation of postoperative complications in gastrointestinal surgeries. The inaccurate and often confusing term, major and minor complications is replaced by the well-standardized Clavien-Dindo classification. Using this standardized system, we analyzed and classified the postoperative complications after curative gastrectomy for gastric cancer. However, due to the limited number of complications in grade or higher, we did not perform specific analysis in each Clavien-Dindo grading. Li et al. [20] showed no significant difference in the 5-year overall survival and disease-specific survival between Clavien-Dindo II group and NC group after total gastrectomy for advanced gastric cancer. However, Duraes et al. [21] showed that complications of minor severity, such as the Clavien-Dindo classification, can adversely affect oncologic outcomes after colorectal cancer resection. In this study, we also found that the increase of Clavien-Dindo grading, including Clavien-Dindo II group, adversely affect the 5-year overall survival and cancer-specific survival after curative gastrectomy for gastric cancer. Because the relationship between the Clavien-Dindo classification and long-term survival outcomes was not linear and uniform, patients with Clavien-Dindo IV complications had worse survival outcomes compared with patients with Clavien-Dindo III complications.
As mentioned above, several previous studies pertaining to gastrointestinal malignancies have indicated that postoperative complications increase cancer-specific mortality rates [8–10,19]. However, the mechanism relating long-term survival of patients with postoperative complications remains unclear. McMillan et al. [22] showed that systemic inflammatory response syndrome is associated with poor survival in patients with curative treatment of colorectal cancer. That is, various mediators of exaggerated systemic inflammatory response caused by postoperative complications may induce release of proinflammatory cytokines [23,24]. The release of proinflammatory cytokines, including tumor necrosis factor-alpha, interleukin (IL)-1beta, IL-6, and IL-18, and oxygen free radicals may alter host defense and promote the motility, invasiveness, and proliferation of residual or implanted tumor cells [25]. Furthermore, cell-mediated immunity, involving cells such as natural killer cells and cytotoxic T lymphocytes, is related to the extent of surgical trauma and tissue damage [26,27]. Systemic infection and sepsis due to postoperative complications may induce a period of immunosuppression, which is enhanced after surgical trauma, and contributes to the proliferation of occult or dormant cancer cells, resulting in decreased survival [28,29]. Consequently, differences in the degree of immune suppression and systemic inflammatory response affect survival according to type of complications. We classified complications as local or SC, and assessed the correlation between type of complications and the 5-year survival in patients undergoing curative gastrectomy for gastric cancer. The observations of our study indicating a low survival rate and high cancer recurrence rate in the LC group due to induced immune suppression and systemic inflammatory response provide additional support for the results discussed above.
Subset analyses indicated no significant differences in cancer-specific survival in stage I, but significant differences between the LC group and the other groups were identified in advanced stage II and III disease. In early gastric cancer, tumor cells are confined to the mucosal layer, and tumor cells are mostly removed by curative radical gastrectomy. However, spreading or spillage of viable exfoliated tumor cells occurs frequently in advanced compared to early gastric cancer [17,30]. When LC develop in advanced gastric cancer, therefore, the resulting effects on the degree of inflammatory immune response and immune suppression may produce a greater number of adverse outcomes with respect to cancer-specific survival and recurrence rates in comparison with NC and SC groups. Tsujimoto et al. [9] also demonstrated a negative impact of infectious complications on gastric cancer recurrence.
Our study had several limitations. First, this was a non-randomized observational study and had a retrospective design. As such studies cannot be based on a randomized controlled experiment due to ethical issues, it is essential to perform a multicenter observational study. However, all consecutive patients undergoing curative gastrectomy for gastric cancer were prospectively included in a multicenter registry, and continuing data collection will improve the generalizability of our findings. Second, with respect to type of complications, we could not investigate the postoperative biological status, such as proinflammatory cytokines, including tumor necrosis factor-alpha, IL-1beta, IL-6, and IL-18, and we could not estimate the degree or duration of immune suppression in this study. Third, grade I complications present any complication not requiring medical or surgical treatment, such as nausea, vomiting, and fever. In fact, since grade I complications were not always considered as complications in clinical practice, data of grade I complications would be unreliable and the overall incidence of grade I complications could be underestimated. Thus, we did not evaluate grade I complications after curative gastrectomy for gastric cancer. Finally, LC delay or prohibit patients from receiving adjuvant chemotherapy to a greater extent than SC. This may account for worse survival in the LC group, but our study could not investigate the percent of patients in each group who delayed or failed to receive adjunctive chemotherapy.
In conclusion, the postoperative occurrence of LC was an independent prognostic factor associated with poor overall and cancer-specific survival, and a high recurrence rate in patients undergoing curative gastrectomy for advanced gastric cancer, especially stage II and III diseases. The negative effect on survival outcomes were also increased with higher Clavien-Dindo grades. These findings suggest that surgeons should implement surgical procedures with careful perioperative care to minimize the occurrence of postoperative complications.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.