INTRODUCTION
Multiple myeloma (MM) also known as plasma cell myeloma is a B-cell malignancy characterized by abnormal proliferation of plasma cells which produce a monoclonal immunoglobulin. Calcium elevation, renal injury, anemia, and bone lesions (CRAB features) are needed for establishing the diagnosis of active myeloma. Skull X-rays and other bony parts in multiple myeloma often show a solitary lytic lesion or multiple punched out areas of bone destruction
When we encounter a patient with CRAB features and multiple osteolytic lesions in skull and other bony parts, we first assume MM. But osteolytic lesions in skull and other bony parts can be found also in metastatic tumors. Here we report a case of breast cancer with osteolytic lesion in multiple bones including skull, which looks more like osteolytic lesion of multiple myeloma.
CASE REPORT
A 60-year-old female was admitted to our emergency room with confused mentality. The patient complained of epigastric and back pain. She did not have any past medical history or social history other than alcohol abuse. On physical examination, epigastric tenderness and pitting edema were observed. Her initial blood pressure was 80/40 mm Hg, heart rate was 85 beat/min, respiratory rate was 25 breath/min, and body temperature was 36.5°C.
Blood test showed white blood cell count of 14,240/mm (neutrophils 83.7%, lymphocytes 11.7%, monocytes 3.1%, and eosinophils 0.4%), hemoglobin level of 11.7 g/dL, total calcium level of 15.3 mg/dL, ionized calcium level of 2.0 mmol/L, total protein level of 5.4 g/dL, albumin level of 2.4 g/dL, uric acid level of 19.6 mg/dL, blood urea nitrogen level of 157.8 mg/dL, creatinine level of 4.2 mg/dL, and C-reactive protein level of 24.6 mg/dL. Urine sample analysis showed one positive dipstick test for protein.
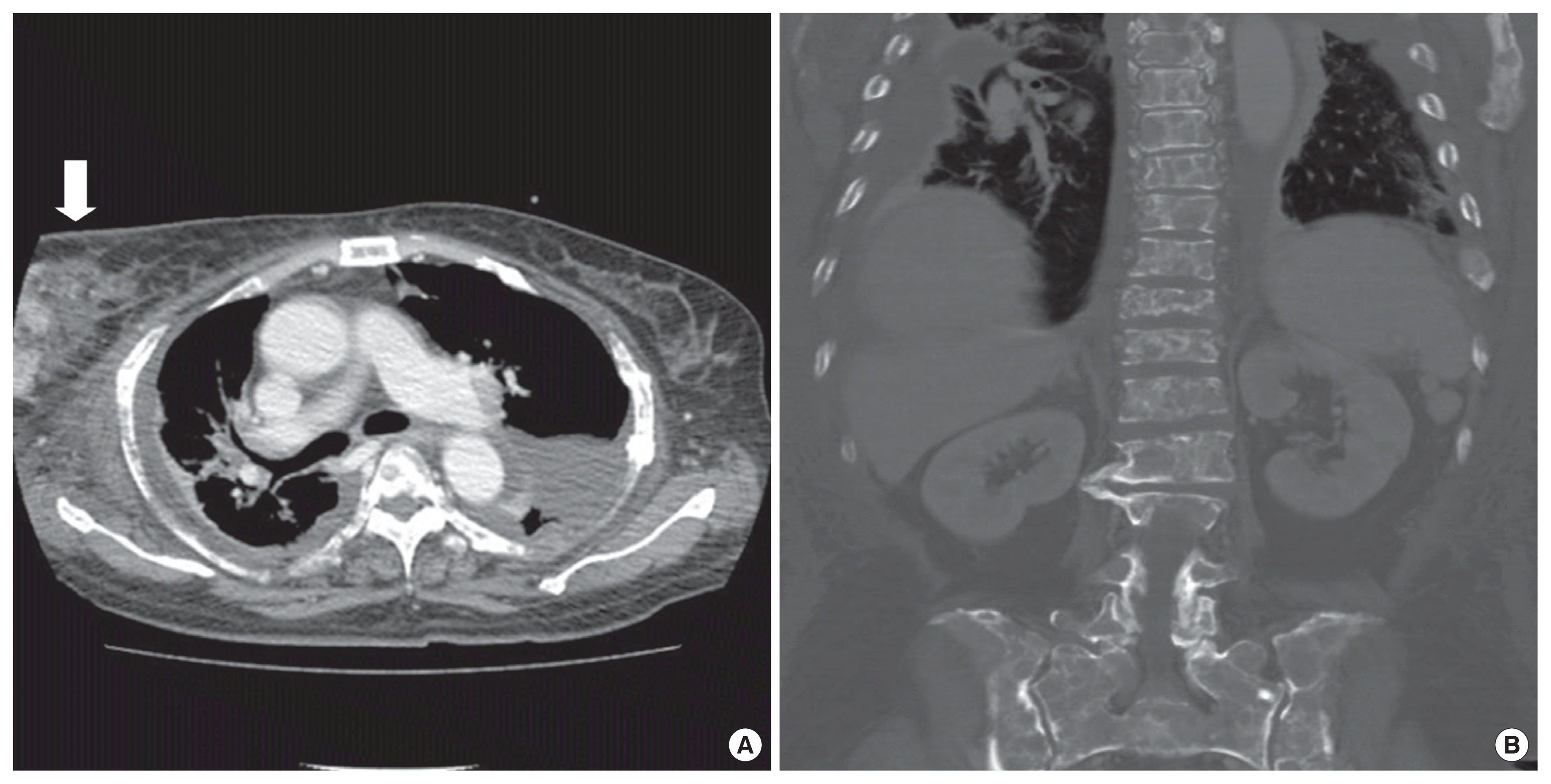
Chest X-ray demonstrated cardiomegaly and pulmonary edema. Multiple osteolytic lesions in the ribs, spine, pelvis, both femurs, and both humeri were detected on both chest and abdominal X-rays. Additional X-rays of the skull and lumbar spine showed punched out lesions (Fig. 1). Brain computed tomography (CT) was performed to identify the cause of altered mentality, and it showed no abnormal signs in the brain parenchyma but multiple osteolytic lesions in the skull (Fig. 2).
Based on the findings of an increased calcium level, kidney failure, anemia, and osteolytic lesions which lead us to suspect MM, further laboratory test was performed. Protein electrophoresis showed no monoclonal peak in serum and urine. Free kappa light chain value was 58.8 mg/L and free lambda light chain value was 44.73 mg/L, and their ratio was within the normal range. Immunofixation electrophoresis showed no diagnostic abnormality and no monoclonal band was detected. β2-microglobulin level of 7.2 mg/L was normal. Bone marrow examination revealed no increase in the number of plasma cells but normocellular bone marrow with involvement of metastatic malignant cells was detected.
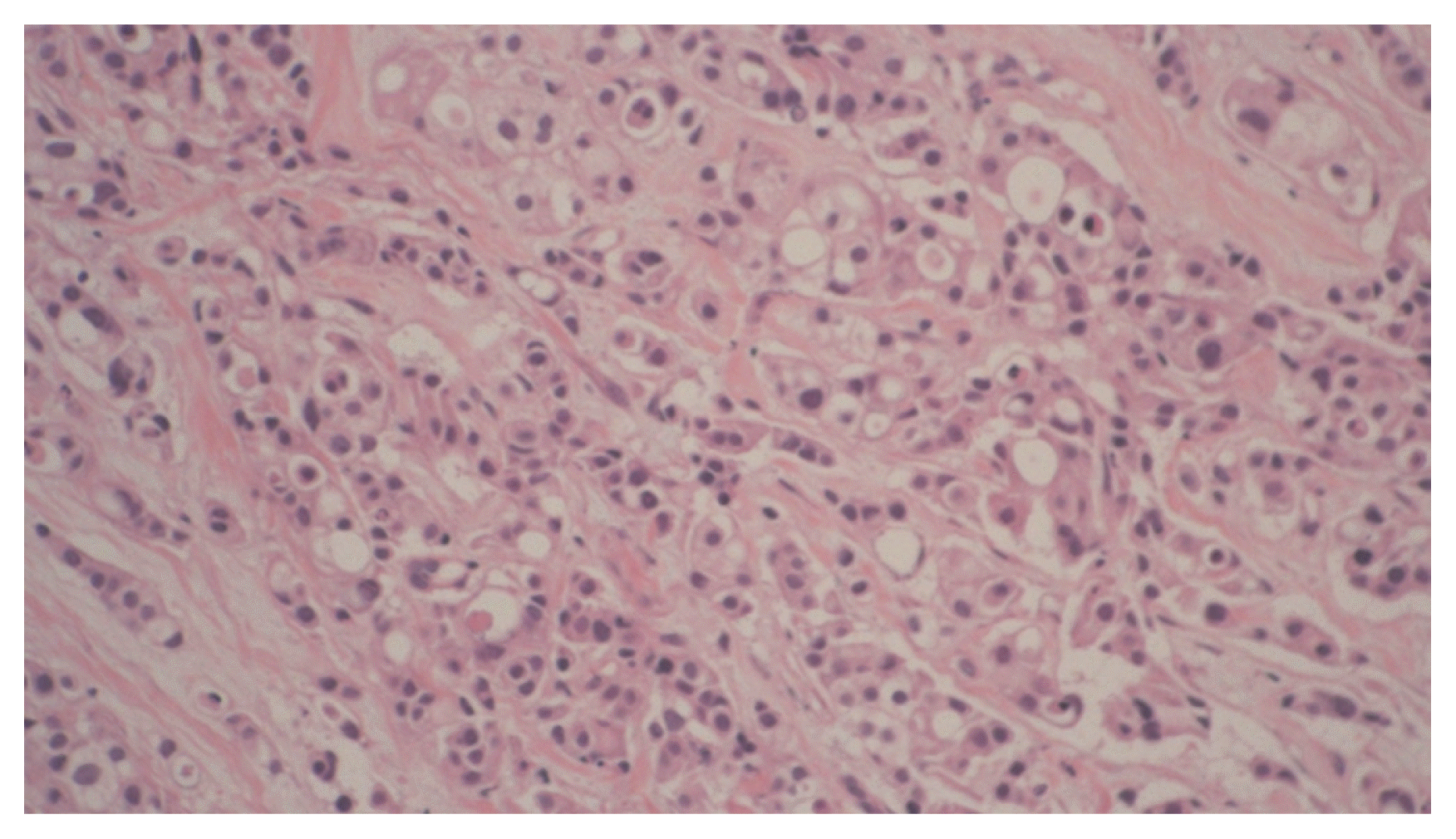
After ruling out MM based on the above results, we thoroughly reperformed the physical examination to detect hidden malignancy, and we detected a palpable mass in the right breast. Tumor markers showed carcinoembryonic antigen level of 367.2 ng/mL, cancer antigen (CA) 19-9 level of 176.7 U/mL, CA 125 level of 384.5 U/ mL, and CA 15-3 level of 300 U/mL, and all these levels were increased. Chest and abdominopelvic CT scans demonstrated a right breast mass with metastasis to multiple axillary and mediastinal lymph nodes, liver, omentum and mesentery, pancreas, and lung (Fig. 3). Breast sonography showed a 5× 2.4× 5.2 cm-sized irregular heterogeneous hypoechoic lesion in the right-upper-outer quadrant (Breast Imaging Reporting and Data System category 5). Eventually, sonography-guided core needle biopsy revealed invasive ductal carcinoma that was estrogen receptor-positive, progesterone receptor-negative, and C-erbB2-three positive (Fig. 4).
After managing initial instability caused by hypercalcemia and acute kidney injury, her mentality recovered to a normal status. Since estrogen receptor and C-erbB2 were positive and she has symptomatic visceral metastasis, we considered human epidermal growth factor receptor 2 and cytotoxic chemotherapy for the initial treatment of breast cancer. However, her performance status was poor and congestive heart failure was diagnosed on the echocardiogram which might worsen with trastuzumab [1,2]. Accordingly, we started only letrozole, an aromatase inhibitor. After we treated the patient with letrozole and analgesics, her pain in the abdomen and bone was reduced and she was discharged to a long-term care facility.
DISCUSSION
When we encounter a patient with multiple osteolytic lesions in the skull, we first assume MM. However, osteolytic lesions in the skull can also be found in metastatic cancers. Skull metastases are found in 5%–6% of patients with advanced systemic cancer. Clinically, they can cause local swelling that is usually painless and they rarely lead to neurological dysfunction [3]. Skull metastases are rarely diagnosed in a clinical setting, but they are frequently found during autopsies. As early as 1889, Paget [4] reported affection of the skull in 36 of 650 post-mortem examinations performed on breast cancer patients. Despite the fact that hematogenous skull metastases can be caused by nearly all types of tumors such as breast, lung, prostate, and kidney malignancies, breast cancer was found to be associated with the highest rate of metastatic skull lesions [5–9].
Bone metastasis may be osteolytic, sclerotic or mixed on radiographs. Osteolytic metastases are most frequently encountered in breast and lung carcinoma. Other rare tumors, such as reticulum cell sarcoma, angiosarcoma, and malignant fibrous histiocytoma, were also reported to show osteolytic bone lesions [10]. Bone modifying agents are an important component of the treatment for bone metastases. Bisphosphonates prevent the complications of breast cancer involving bones, such as fractures, spinal cord compression, and hypercalcemia of malignancy. However, intravenous bisphosphonates are generally not recommended in patients with creatinine clearance less than 30 mL/min because of the risk of renal toxicity [11]. Since the patient had kidney dysfunction, the use of bisphosphonates was contraindicated [12].
MM is a B-cell malignancy characterized by abnormal proliferation of plasma cells which produce a monoclonal immunoglobulin. High blood calcium levels, renal injury, anemia, and bone lesions are needed for establishing the diagnosis of active myeloma. Skull X-rays in multiple myeloma often show a solitary lytic lesion or multiple punched out areas of bone destruction [13]. The patient described in this report had high blood calcium levels, renal injury, anemia and osteolytic bone lesions, which complied with the clinical features of MM. However, the monoclonal component was not detected in protein electrophoresis and bone marrow examination did not prove the increase in the number of plasma cells. According to the International Myeloma Working Group diagnostic criteria for MM, myeloma was ruled out in this patient [14]. Then further evaluation for a solid tumor was performed and breast cancer was finally diagnosed.
So far only one study has reported a case of a patient with multiple osteolytic lesions in the axial skeleton, ribs, skull, pelvis, both femurs, and both humeri [15]. The patient was admitted with a provisional diagnosis of multiple myeloma, but no evidence of paraprotein was detected in the blood or urine. Bone marrow biopsy revealed carcinoma cells that were positive for CA 15-3, estrogen receptor positive (51%–75%), and HER-2 (3+). As a result, breast cancer with bone metastases was diagnosed. In this report, calcium level and the results of kidney function were normal, which were abnormal in our case, and hence, our case had more similarity with myeloma.
Osteolytic skull metastasis is one of the typical manifestations of MM. However, we should also consider the possibility of metastatic carcinoma especially breast cancer as well as MM in the differential diagnosis of osteolytic skull metastasis.