Adequacies of lymphadenectomy range for gastric cancer according to the second and third/fourth Japanese gastric cancer treatment guidelines
Article information
Abstract
Purpose
This study evaluated the adequacies of lymph node (LN) dissection according to the second version (determined by tumor location) or third/fourth version (determined by surgery extent) of the Japanese gastric cancer treatment guidelines.
Methods
Prospectively collected data of 3,948 gastric cancer patients who underwent gastrectomy were analyzed. The prevalence of LN metastasis and 5-year survival were analyzed according to tumor invasion depth and tumor location. In early gastric cancer (EGC), the frequency of LNs were evaluated. In advanced gastric cancer (AGC), the frequency of LN metastasis and the 5-year survival rate of patients with positive LN were evaluated.
Results
For lower-third EGC, the positive rates for the #1 and #4sb were 0.93% and 0%. For upper-third EGC, the positive rates for #4d, #5, #6, and #11p were 0.3%, 0%, 0.76%, and 1.22%. For lower-third AGC, the positive rates for #4sb and #14v were 2.48% and 7.64%, and the 5-year survival rates were 69.2% and 12.5%, respectively. For upper-third AGC, the positive rates for #5, #6, and #12a were 2.33%, 2.57%, and 2.03%, and the 5-year survival rates were 21.8%, 64.3%, and 0%, respectively.
Conclusion
According to our analysis, in EGC, LN dissection in second edition seems more suitable, however LN dissection in #11p would be mandatory in upper third EGC. In AGC, LN dissection in third/fourth edition seems more suitable in terms of frequency of LN metastasis and survival rate.
INTRODUCTION
Surgery is the main curative treatment for gastric cancer [1,2], and prognosis can be predicted by perigastric lymph node (LN) metastasis and depth of invasion as part of the TNM staging [3,4]. Previous studies have revealed that complications, survival, and recurrence may vary according to the range of LN dissection [5–8]. For example, the Dutch trial revealed that D2 dissection decreased the rates of cancer-related mortality and local recurrence (vs. D1 dissection), but increased the rates of operative mortality and complications [5,6]. In contrast, the Japan Clinical Oncology Group trial compared D2 and para-aortic dissection (D3), and failed to detect differences in recurrence-free survival, although D3 dissection was associated with more complications, bleeding, and operative time [7]. Other studies have also indicated that more radical dissection is not beneficial for improving the prognosis of patients with gastric cancer [8,9]. Thus, it is important to determine the appropriate range for LN dissection in cases of gastric cancer.
The Japanese Gastric Cancer Association (JGCA) has proposed recommendation for LN dissection in cases of gastric cancer [10–12], which generally involves D1 or D1+ dissection for early gastric cancer (EGC) and D2 dissection for advanced gastric cancer (AGC). However, the second and third/fourth editions of the JGCA treatment guidelines have minor differences, as the dissection range was determined using tumor location in the second edition, compared to gastrectomy extent in the third/fourth edition. Although a simple comparison is difficult, as the extent of gastrectomy depends on the tumor location, the frequencies of LN metastasis for specific tumor locations may help determine the appropriate extent of LN dissection [13]. Therefore, the present study aimed to evaluate the adequacies of LN dissection extent based on the second and third/fourth editions of the JGCA guidelines, using the rates of LN metastasis and survival for each positive LN station.
METHODS
This retrospective study examined prospectively collected data from 3,948 patients who underwent gastrectomy for gastric cancer at Seoul National University Hospital between January 2011 and December 2015. The study’s protocol was approved by the Institutional Review Board (IRB no. 1707-068-869), and the data were collected from electronic medical records while protecting patient anonymity.
We followed American Joint Committee on Cancer seventh edition for gastric cancer staging. The grouping of LNs was performed by the attending surgeon immediately after the gastrectomy, and the LN stations were classified according to the JGCA system [14]. Tumor location was defined as the upper-third, middle-third, or lower-third of the stomach; cases that involved the entire stomach were excluded. Surgery after endoscopic submucosal dissection was performed for cases with a possibility of LN metastasis [15], and invasion to other organs was treated in an attempt to improve the patient’s prognosis. The tumor’s depth was classified as EGC or AGC, based on the pathological results.
To compare the second and third/fourth editions of the JGCA guidelines, upper-third lesions were assumed to have been treated using total gastrectomy, while middle-third and lower-third lesions were assumed to have been treated using distal gastrectomy. Table 1 summarizes the LN dissection extents from the two guidelines, based on the assumption that D1+β (second edition) or D1+ (third/fourth edition) were the standard extents for EGC, with D2 being the standard extent for AGC. The patients’ data were also examined to determine the frequency of LN metastasis in EGC cases, as well as survival and specific LN metastases in AGC cases.
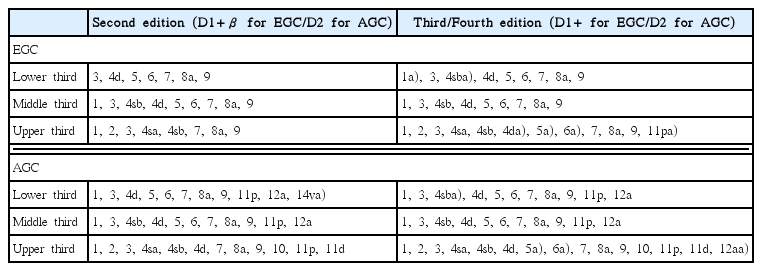
Comparing lymph node dissection between the second and third/fourth editions according to tumor location
There was no difference between the second and third/fourth versions for middle-third gastric cancer. Thus, we only investigated gastric cancers in the lower-third and upper-third. For each station, the frequency of LN metastasis and survival rate were evaluated, with frequency being defined as the percentage of positive LNs among the resected LNs, which is a useful prognostic factor [4]. Stations were considered mandatory for dissection (1) if the frequency of LN metastasis was = 1% in EGC cases or (2) patients with positive LNs and AGC had a 5-year survival rate of = 20%. In some papers investigating the frequency of LN metastasis in EGC, the incidence of perigastric lymph nodes was reported to be 1.2%–7%, suggesting that more than 1% of LN metastasis woule be suitable for mandatory dissection [16–19]. Also in our hospital data, 5-year survival after curative resection of gastric cancer showed that stage IIIa was 53.9%, IIIb was 36.5%, and IV was 23.9%. So, we think that 5-year survival showed over 20% than the dissection would be meaningful [1].
Data were analyzed using IBM SPSS software ver. 21 (IBM Corp., Armonk, NY, USA) and Excel software (version 2013). Data were reported as number (%) and compared using frequency analysis. Survival outcomes were compared using the Kaplan-Meier method.
RESULTS
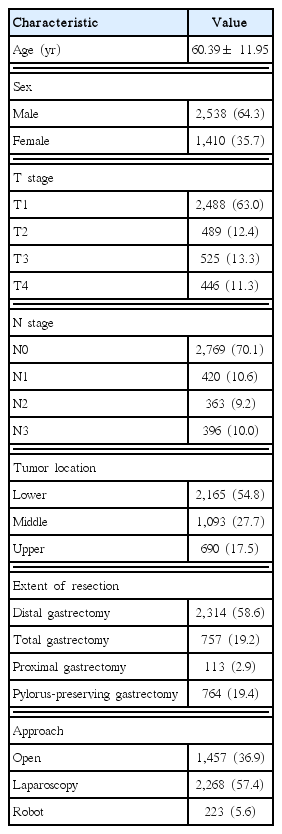
Demographic characteristics
Table 2 shows the patients’ demographic characteristics. The mean age was 60.39 ± 11.95 years. Most patients were men (64.3%, n= 2,538), had EGC (63.0%, n= 2,488), and did not have LN metastasis (70.1%, n= 2,769). The cases with LN metastasis involved N1 metastasis (10.6%, n= 420), N2 metastasis (9.2%, n= 363), and N3 metastasis (10.0%, n = 396). The tumor locations were upper-third (17.5%, n = 690), middle-third (27.7%, n = 1,093), and lower-third (54.8%, n= 2,165). Most patients underwent distal gastrectomy (58.6%, n= 2,314) or total gastrectomy (19.2%, n= 757). Smaller proportions of patients underwent proximal gastrectomy (2.9%, n = 113) or pylorus-preserving gastrectomy (19.4%, n = 764).
Adequacy of LN dissection for EGC
Fig. 1 shows the frequencies of LN metastasis from lower-third or upper-third EGC. For lower-third EGC, stations 1 and 4sb were added to the third/fourth edition and had metastasis rates of 0.93% and 0%, respectively. For upper-third EGC, stations 4d, 5, 6, and 11p were added to the third/fourth edition as D1+LN. Only 11p had a metastasis rate of > 1% (1.22%, 4/328), with lower rates for 4d (0.3%, 1/332), 5 (0%, 0/226), and 6 (0.76%, 2/262).
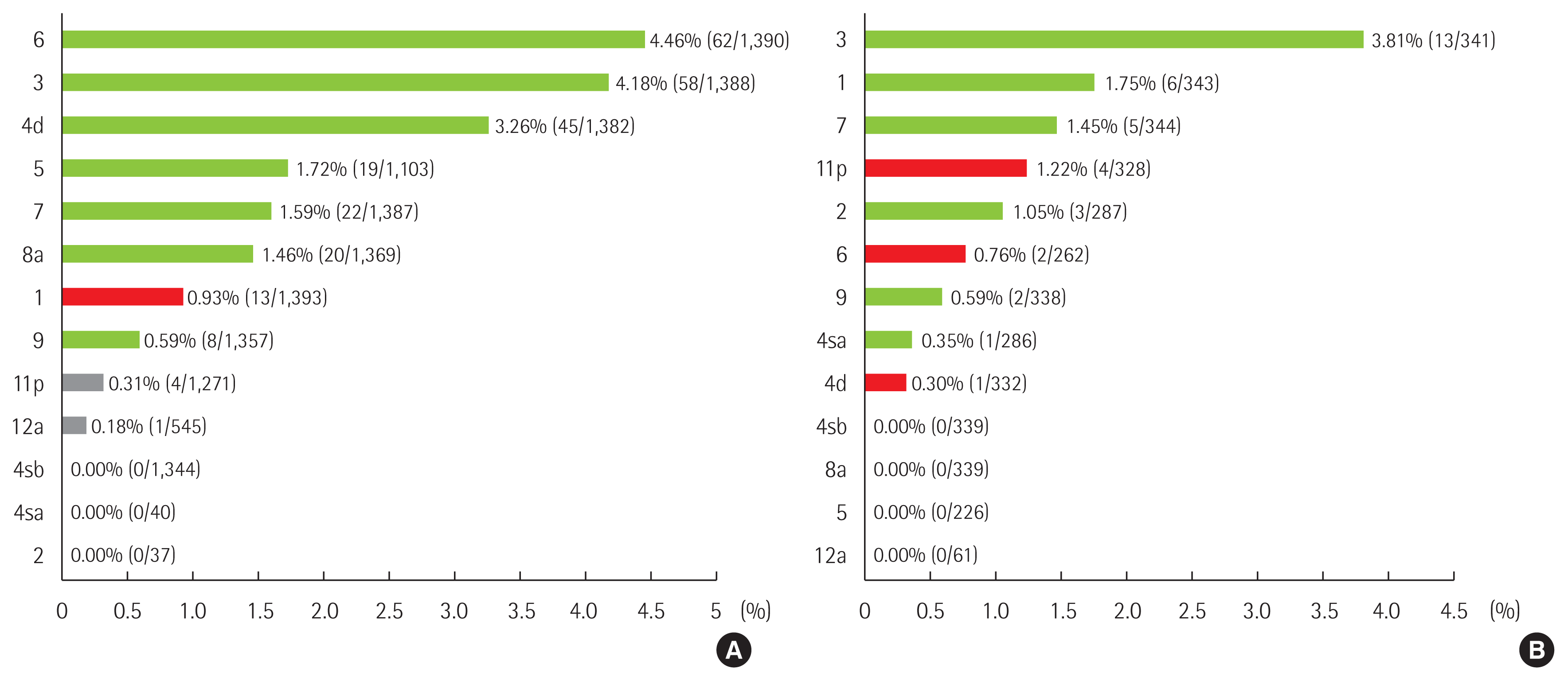
Frequency of lymph node (LN) metastasis in early gastric cancer (EGC). (A) LN metastasis (%) in the lower third EGC. (B) LN metastasis (%) in the upper third EGC. Green bar means LN station within D1+ in the second and third/fourth editions. Red bar means LN station beyond D1+ in the second edition, but within D1+ in the third/fourth edition. Grey bar means LN station beyond D1+ in the second and third/fourth editions.
Adequacy of LN dissection for AGC
Fig. 2 shows the frequencies of LN metastasis and survival for lower-third or upper-third AGC. For lower-third AGC, station 4sb was added and 14v was removed from the third/fourth edition, and had metastasis rates of 2.48% (18/726) and 7.64% (12/157), respectively. Patients with 4sb metastasis had a 5-year survival rate of 69.2%, while patients with 14v metastasis had a 5-year survival rate of just 12.5%. For upper-third AGC, stations 5, 6, and 12a were added to the third/fourth edition, and had metastasis rates of 2.33%, 2.57%, and 2.03%, respectively. Patients with station 5 metastasis had a 5-year survival rate of 21.8%. Among the eight patients with station 6 metastasis, the 5-year survival rate was 64.3%. All four patients who had station 12a metastasis died within 3 years.
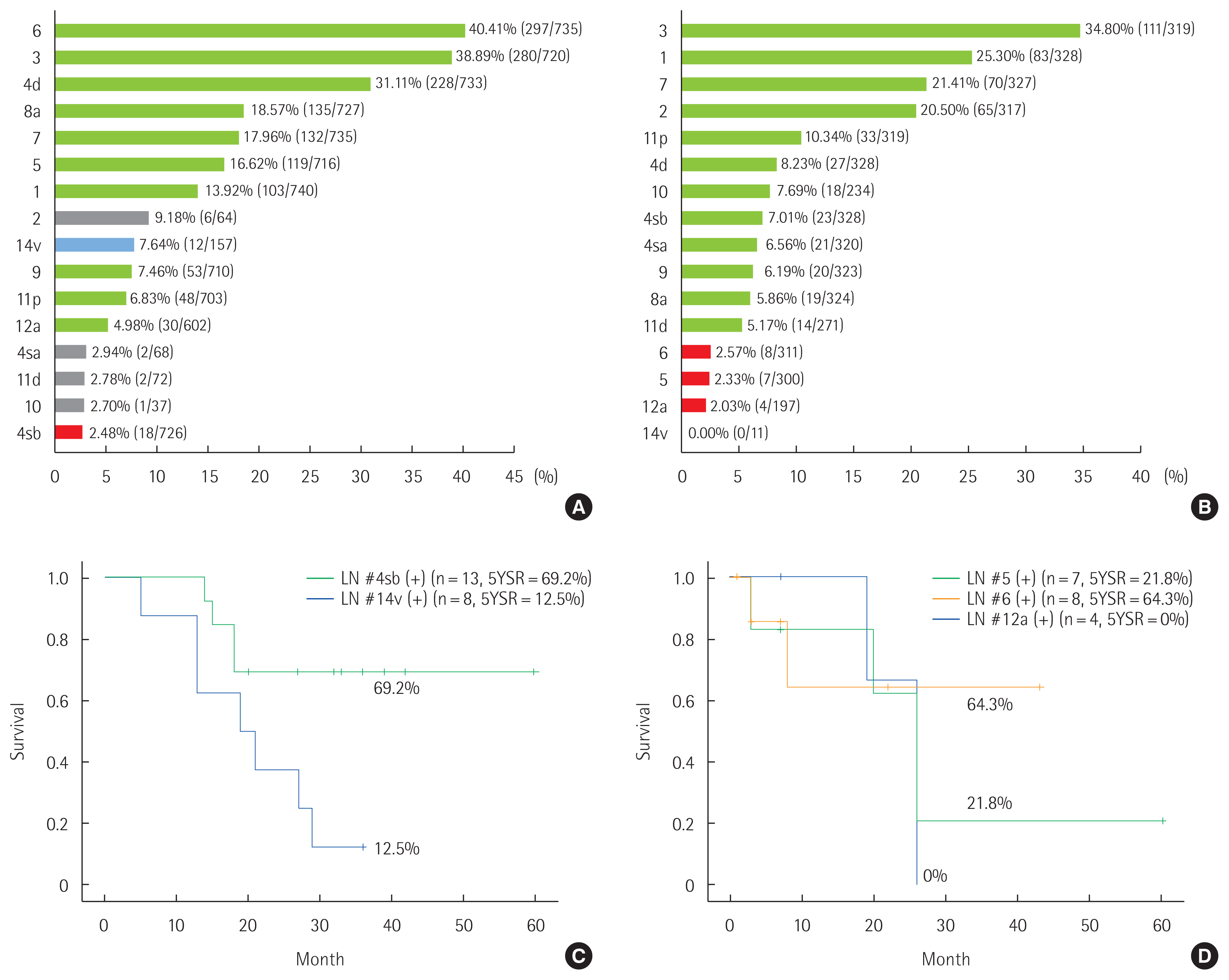
Frequency of lymph node (LN) metastasis in advanced gastric cancer (AGC) and survival according to specific metastases. (A) LN metastasis (%) in the lower third AGC. (B) LN metastasis (%) in the upper third AGC. (C) Survival curve in the lower third AGC. (D) Survival curve in the upper third AGC. Green bar means LN station within D2 in the second and third/fourth editions. Red bar means LN station beyond D2 in the second edition, but within D2 in the third/fourth edition. Grey bar means LN station beyond D2 in the second and third/fourth editions. Blue bar means LN station within D2 in the second edition, but beyond D2 in the third/fourth edition. 5YSR, 5-year survival rates.
DISCUSSION
Gastric cancer frequently leads to LN metastasis, which is the most important prognostic factor for gastric cancer [4,20], and it is important to understand the adequacy of LN dissections. Our center and many other Korean hospitals follow the JGCA recommendations for treating gastric cancer. Thus, it was important for us to examine the changes in the second and third/fourth editions of the JGCA guidelines, and to question whether location-based dissection would be more appropriate.
For lower-third EGC, dissection of stations 1 and 4sb is not mandatory, because the metastasis frequencies were < 1%. However, for AGC, the metastasis rates were > 1% for stations 14v and 4sb, which indicates that their dissection should be considered mandatory in terms of frequency. Furthermore, 11p dissection for upper-third EGC was added in the third/fourth edition, and had a metastasis rate of > 1%, which indicates that it should be considered mandatory. Nevertheless, the 4 patients with 11p metastasis from upper-third EGC survived throughout the follow-up period. Stations 5 and 6 had metastasis rates of < 1% and should not be considered mandatory for dissection.
We considered the survival of all patients with lower-third AGC because they had a metastasis rate of < 1%. In that analysis, the 5-year survival rates were 69.2% for 4sb and 12.5% for 14v (most patients with 14v metastasis died, and the one surviving patient did not complete the 5-year follow-up). These results indicate that 4sb dissection should be considered mandatory for lower-third AGC, although 14v dissection is less important, which favors the third/fourth edition of the JGCA guidelines. Interestingly, the four patients with 11p metastasis from upper-third EGC survived, which suggests that 11p dissection should be considered mandatory based on the third/fourth edition of the JGCA guidelines. However, among AGC cases, the 5-year survival rates were 21.8% for station 5, 64.3% for station 6, and 0% for station 12a, which suggests that 12a dissection is not mandatory based on the second edition, despite the relatively high metastasis rate. This may be because all four patients with 12a metastasis died because of recurrence within 3 years after surgery. A low 5-year survival rate was observed for patients with station 5 metastasis, although one of the five patients survived to 5 years without recurrence, which suggests that this is a mandatory dissection.
The present study has several limitations. There are too small patients in each survival analyses ranging 4 to 13 patients. Although this data showed the trend of survival rate but it does not have statistical significance. To see the survival, multivariable factors should be considered but excluding some specific lymph nodes, we didn’t analyze all the factors influencing the survival could not be investigated. And about 20% of the patients were underwent limited gastrectomy with limited LN dissection such as proximal gastrectomy or pylorus preserving gastrectomy, and it that might affect the frequency of LN mestastasis in EGC.
In conclusion, we assessed the adequacies of LN dissection based on the JGCA guidelines, which had several changes between the second and third/fourth editions, especially for AGC. In cases of lower-third EGC, dissection of stations 1 and 4sb is not mandatory, because of their low metastasis rates. However, in cases of lower-third AGC, it is reasonable to consider 4sb mandatory for dissection and to consider omitting 14v dissection. In cases of upper-third EGC, dissection of stations 4d, 5, and 6 does not appear to be mandatory, which favors the second edition, although 11p dissection is mandatory, which favors the third/fourth edition. In cases of upper-third AGC, dissection of 12a does not appear to be mandatory, while dissection of stations 5 and 6 is mandatory, which favors the third/fourth edition.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.