폐쇄성 대장암의 수술 전 처치로써 내시경적 자가팽창 스텐트 삽입술의 종양학적 안정성
Oncologic safety of self-expanded metal stent insertion as a bridge to elective surgery in malignant colorectal obstruction
Article information
Trans Abstract
Purpose
Colorectal obstruction develops most frequently by carcinoma, and 7%–30% of these colorectal carcinomas are acute cases. The oncologic safety of self-expanding metal stent (SEMS) insertion as a bridge to surgery has not yet been established. Thus, we investigated the oncologic safety of SEMS insertion as a bridge to surgery in patients with obstructive colorectal cancer.
Methods
This retrospective had 56 patients enrolled requiring emergency management for obstructive colorectal cancer at stage II or III, who had undergone curative surgery between July 2008 and June 2011. These subjects were divided into two groups: patients who had undergone emergency surgery without SEMS insertion (non-stent group) and those who had undergone elective surgery after preoperative decompression with SEMS insertion (stent group). The two groups were compared for clinicopathologic characteristics, postoperative complications, and survival rate.
Results
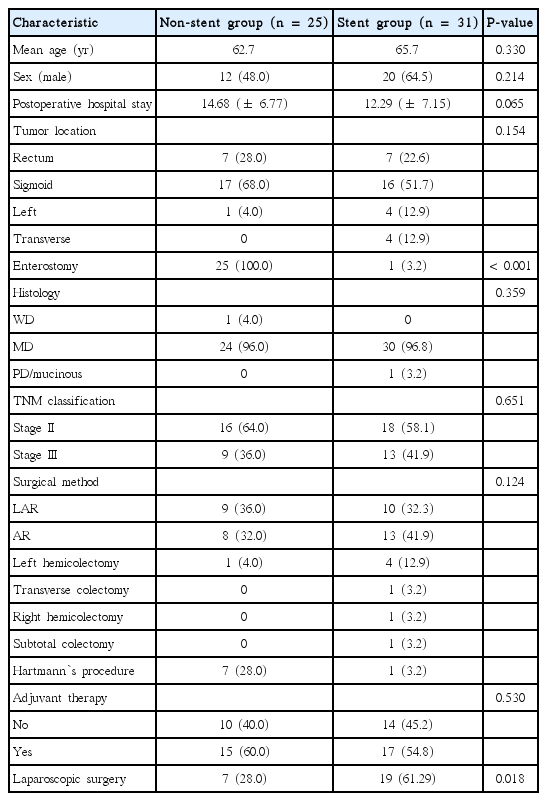
Enterostomy was performed in 25 patients (100.0%) in the non-stent group and 1 patient (3.2%) in the stent group; laparoscopic surgery was carried out in 7 patients (28.0%) in the non-stent group and 19 patients (61.29%) in the stent group, each showing statistically significant differences. There was no statistically significant difference in postoperative complications and 5-year disease-free survival rate (72% vs. 74.19%, P=0.87, respectively).
Conclusion
In treatment of malignant colorectal obstruction, elective operation after stent insertion had similar oncologic outcomes compared with emergency operation. Preoperative stent insertion not only lowers the incidence of enterostomy but also makes laparoscopic surgery possible, thereby enhancing patients’ quality of life. Therefore, preoperative stent insertion is a useful method that may replace emergency surgery in treatment of malignant colorectal obstruction.
INTRODUCTION
Colorectal cancer is the third most common cancer in Korea. It is the second most common cancer in males and the third most common in females [1]. A malignant colorectal obstruction develops most frequently by carcinoma, and 7%–30% of these colorectal carcinomas are acute cases [2]. Various approaches have been attempted by surgeons of diverse inclinations, and conventionally include emergency colostomy, Hartmann’s procedure, and subtotal colectomy.
Owing to unfavorable systemic conditions (such as dehydration and electrolyte imbalance), as well as older age, notwithstanding advances for perioperative management, emergency surgery in patients with malignant colorectal obstruction is known to be associated with a higher morbidity or mortality rate than primary surgery [3-6]. Furthermore, only 60% of patients who undergo Hartmann’s procedure receive colostomy reversal [3]; an adequate assessment of a concurrent lesion may not be carried out in cases of emergency surgery. Recently, self-expanding metal stent (SEMS) insertion has been performed for preoperative decompression in obstructive lesions without emergency surgery. This allows colonic irrigation and improved systemic conditions before undertaking primary surgery. Some studies have reported on this issue [4,5,7-9]. The oncologic safety of SEMS insertion as a bridge to surgery has not yet been established. Thus, we investigated oncologic safety of SEMS insertion as a bridge to surgery in patients with obstructive colorectal cancer.
METHODS
The current retrospective study included 56 patients requiring emergency management for obstructive colorectal cancer at stage II or III, who had undergone curative surgery at the division of colorectal surgery at the Department of General Surgery, Inje University Busan Paik Hospital from July 2008 to June 2011. These subjects were divided into two groups: patients who had undergone emergency surgery without SEMS insertion (non-stent group, 25 patients), and those who had undergone primary surgery after preoperative decompression with SEMS insertion (stent group, 31 patients).
We performed emergency surgery in the following cases: (1) If patients have unstable vital signs. (2) If immediate bowel decompression is required in radiologic imaging studies and physical examination. (3) If the SEMS insertion failed.
The remaining cases underwent SEMS insertion as a bridge to elective surgery. The patients’ age, gender, location of lesion, histologic staging, surgical method, and hospital duration after surgery, as well as postoperative complications, were examined from their medical records. The diagnosis of malignant colorectal obstruction was confirmed by the suspicion of an obstructive lesion in the abdominal X-ray and computed tomography, having clinical symptoms of nausea, vomiting, abdominal pain and abdominal distension. Curative resection was defined as having no residual cancerous tissues visually confirmed during surgery and no malignant tissues in the surface margin of a resected specimen, as verified in the histopathologic results. The locations of the lesions were classified based on surgical findings, while tumor, node, metastasis (TNM) staging was based on the histopathologic diagnosis after surgery.
SEMS insertion
Uncovered colonic SEMS (S&G Medical, Seoul, Korea) insertion was performed under fluoroscopic guidance. After identification of the obstructive lesion, a guide wire was passed through the narrowed lumen. The delivery system was inserted over the guide wire so that the stent was positioned in the middle of the narrowed segment, and the stent was deployed. Persistent obstructive symptoms for 48 hours after SEMS insertion were considered a functional failure, and additional SEMS insertion or emergent colostomy was performed. After SEMS insertion, the time (7–9 days after SEMS insertion) for elective surgery was decided by confirming the disappearance of intestinal obstruction and edema through assessment of the patient’s systemic condition and simple abdominal X-ray results. Two liters of Colyte-F powder were prescribed 1 or 2 days before surgery for mechanical colonic irrigation in all patients who underwent elective surgery.
Follow-up observations
Patients were followed up every 3 months for the first 2 years after surgery and every 6 months thereafter for 3 years. The patients’ medical history, physical examination and serum carcinoembryonic antigen levels were recorded at each follow-up visit. Chest X-ray and abdominopelvic computed tomographic scans were performed to assess the efficacy of chemotherapy every 3 months, and every 6 months after the completion of chemotherapy.
A colonoscopy was performed annually. Recurrence was identified by imaging studies and colonoscopy, and confirmed by colonoscopic or percutaneous biopsy. Radiologically identified tumor growth within the previous surgical field would indicate recurrence, particularly when histological confirmation was not possible.
Statistical analysis
All data were presented by mean±standard deviation for continuous variables and as count and percentage for discrete variables. Clinical characteristics were analyzed between with and without SEMS insertion groups using chi-square test or Fisher’s exact test for discrete variable and using Wilcoxon rank sum test for continuous variables. The Kaplan-Meier method with log-rank test was conducted for comparing the survival rates. Independent prognostic factors associated with the survival time were studied using a Cox regression analysis. A P-value less than 0.05 was considered to indicate statistical significance. All statistical analysis was performing using SAS ver 9.3 (SAS Institute Inc., Cary, NC, USA).
RESULTS
Clinical and pathological characteristics
Curative resection was performed on 56 patients with obstructive colorectal cancer; 25 of these patients underwent emergency surgery without SEMS insertion (non-stent group), and 31 underwent SEMS insertion as a bridge to elective surgery (stent group). No statistically significant difference was found in gender, age, tumor location, histopathological differentiation, stage, and adjuvant chemotherapy between these two groups. Enterostomy was performed for 25 patients (100%) in the non-stent group and 1 patient (3.2%) in the stent group, showing a statistically significant difference (P<0.001). Among 31 patients with SEMS insertion in our study, 1 patient (3.2%) with distal transverse colon cancer who had developed colonic perforation after SEMS insertion underwent laparotomy followed by Hartmann’s procedure. There was also statistically significant difzference between the non-stent group (7 patients, 28.0%) and stent group (19 patients, 61.29%) in which laparoscopic surgery was carried out (P=0.018). Furthermore, there was a tendency of shorter postoperative hospital stay in the stent group (12.29 days) than the non-stent group (14.68 days) (P=0.065) (Table 1).
Postoperative complications
There were 15 cases of postoperative complications in this study: 7 patients (28.0%) in the non-stent group and 8 patients (25.8%) in the stent group. Neither group had anastomotic leakage; wound infection (three vs. four patients, respectively; P=0.919) and ileus (four vs. four patients; P=0.646) showed no statistically significant differences between these two groups (Table 2).
Local recurrence was not observed in the non-stent group, while there were two recurrences (6.4%) in the stent group; distant metastasis was found in six cases (24.0%) in the stent group and seven cases (22.6%) in the non-stent group, showing no statistically significant difference (P=0.214) (Table 2).
Analysis of survival rates and prognostic factors
In the univariate analysis, the prognostic factor for disease free survival (DFS) was age (P=0.021) (Table 3).
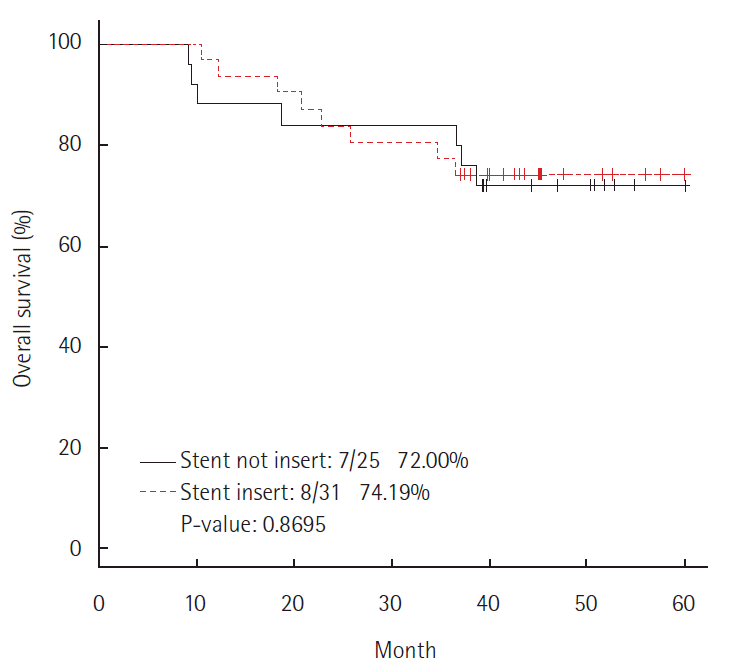
There was no statistically significant difference in the 5-year survival rate between the non-stent group (72%) and stent group (74.19%) (P=0.869) (Fig. 1). Furthermore, there was no significant difference in the 5-year DFS rate between the non-stent group (71.25%) and stent group (70.97%) (P=0.670) (Fig. 2).
DISCUSSION
Some cases with endoscopic colonic stent placement for malignant rectal obstruction as a palliative treatment were reported in the early 1990s [10,11]. Tejero et al. [12] reported in 1994 that SEMS had been used as a bridge to surgery for patients with colonic obstruction. Thereafter, endoscopic colonic stent placement has been performed for colonic irrigation as a preoperative palliative approach before undertaking primary surgery in malignant colorectal obstruction. Endoscopic stent placement has the following advantages: (1) it reduces the risk of emergency surgery, (2) it allows a decision on tumor staging before surgery, (3) one-stage surgery is possible after preoperative colonic irrigation, and (4) it can be utilized as a palliative treatment in either unresectable cases of multiple metastases or cases with high surgical risks [4,7,13-15].
The Dutch stent-in study group [16] reported on patients with acute left-sided malignant colonic obstruction who received either stent placement or emergency surgery. No difference in the 30-day mortality, overall mortality or morbidity was observed between these two groups. The early postoperative stoma rate of the stent placement group was lower than that of the other group. However, it was reported that there was no difference in the stoma rate between these two groups at the end of follow-up observations for 6 months or more. Although this study was prematurely terminated due to a higher 30-day morbidity in stent placement group, they asserted that stent placement in patients with acute left-sided malignant colonic obstruction might be used as an alternative treatment, but clinicians should pay attention to tumor spreading caused by perforation. As mentioned above, stent placement has advantages of providing a palliative treatment and enabling one-stage surgery. However, reports have shown that direct manipulation of the tumor during surgery may adversely affect patients oncologically. There are debates concerning the oncopathologic effect of stent expansion on tumor dissemination after stent placement [17,18]. Turnbull et al. [19] insisted, through the introduction of the notouch isolation technique in 1967, that tumor manipulations during surgery might spread and cause seeding of these tumor cells. The conventional theory by Turnbull et al. [19] affirms that physiological tumor compression by stent placement clearly violates the no-touch isolation technique. Maruthachalam et al. [20] reported that the cytokeratin 20 mRAN expression following stent insertion significantly increased. They suggested that endoscopic stent insertion results in dissemination of cancer cells into the peripheral circulation.
There are various reports of the oncologic effect of stent placement on obstructive colorectal cancer. Saida et al. [5] reported that there was no difference in the 3-year survival rate (48% vs. 50%) or the 5-year survival rate (40% vs. 44%) between the stent placement group and emergency surgery group, respectively. Tung et al. [8] insisted that there was no statistically significant difference in the 5-year survival rate (57.1% vs. 42.8%) between the stent placement group and emergency surgery group in patients with stage II and III disease who had undergone curative resection, respectively, and that stent placement would be oncologically safe in obstructive colon cancer. In our study, the oncologic results from 31 patients who underwent stent placement and 25 patients who underwent emergency surgery, among patients with malignant colorectal obstruction requiring an emergency treatment, were examined. There was no difference in the 5-year survival rate (P=0.869) or 5-year DFS rate (P=0.670) between these two groups. These survival rates were not different from those of other reports. The oncologic safety of stent placement was also substantiated in this study. Kim et al. [21] investigated the stent placement group of patients with obstructive colon cancer and the group of patients with non-obstructive colon cancer who underwent primary surgery. The 5-year survival rate of the stent placement group (48.3%) was lower than that of the primary surgery group (75.5%) (P=0.024). This result was different from those of various other previous reports. However, these authors raised the question of whether this was due to the obstruction, which would be a risk factor for the recurrence of colon cancer.
Preoperative colonic irrigation decreases fecal bacteria, reducing infection in the surgical site. It is known that this procedure allows easy bowel manipulation during surgery and may prevent unexpected fecal spillage [22-24]. However, the Cochrane review [25] reported from the comparisons between the group of patients with colonic irrigation and group of patients without colonic irrigation, that there were no differences in anastomotic leakage and wound infection; however, many surgeons perform largely preoperative colorectal irrigation. In our study, poor preoperative colonic irrigation was shown in all patients (100%) in the non-stent group. Among the patients in the stent placement group, there were 16 cases (51.6%) with good preoperative colonic irrigation (well-group) and merely 4 cases (12.9%) with poor preoperative colonic irrigation (poor-group). Thus, stent placement is significant in that it enables one-stage surgery with primary anastomosis after preoperative colonic irrigation.
Khot et al. [14] reported, from their systemic review associated with stent placement, that the technical success rate was 91.9%, and the clinical success rate was 71.7%, in patients who underwent stent placement followed by primary surgery. They reported that the important complications were intestinal perforation, hemorrhage, stent movement and stenosis. Many studies also conducted investigations on short-term complications, such as the validity and safety of stent placement [7,15,26]. Among 31 patients with SEMS insertion in our study, 1 patient (3.2%) with distal transverse colon cancer who had developed a colonic perforation after SEMS insertion underwent laparotomy followed by Hartmann’s procedure. Lee et al. [27] investigated 130 patients who underwent stent placement among patients with obstructive colon cancer. They found that eight patients (6.2%) developed perforation and reported that the mean stenosis angle of patients with perforation was more acute than that of patients without perforation. The splenic flexure is the area in which the mean stenosis angle is the most acute in the colon. In our study, stent-induced perforation occurred in the area of distal transverse colon cancer. It is known that tumor perforation is a high risk factor in the treatment of colon cancer. In our study, liver metastasis and peritoneal carcinomatosis developed a year after surgery in patients with perforation. Thus, stent placement might be performed selectively in patients with malignant colonic obstruction.
Hong and Kim [28] reported in their systemic review associated with stent placement that not only stoma creation was significantly lower in the SEMS group than in the surgery group (P=0.03), but also primary anastomosis was achieved significantly more frequently in the SEMS group than in the surgery group (P<0.001). Another systemic review also reported that the primary anastomosis rate was significantly higher in the stent group (64.9%) compared with the emergency surgery group (55%) (P=0.003) [29]. In our study, the primary anastomosis rate was higher in the stent group (96.8%) than in the non-stent group (72%). Furthermore, the enterostomy rate was significantly lower in the stent group (3.2%) than in the non-stent group (100%) (P<0.001). Thus, the SEMS insertion as a bridge to elective surgery may provide surgical advantages, such as higher primary anastomosis rate and lower enterostomy rate.
The Dutch stent-in study group [16] reported that 5 of 47 patients with stent placement developed anastomotic leakage. Cirocchi et al. [29] reported that there was no statistically significant difference in anastomotic leakage rate (P=0.35), intra-abdominal abscess rate (P=0.97) and wound infection (P=0.17) between the two groups. In our study, anastomosis was performed without leakage for all of the patients who underwent stent placement, except for one patient who developed stent-induced bowel perforation. Furthermore, there were no significant differences in postoperative wound infection or postoperative ileus between both groups.
In conclusion, in treatment of malignant colorectal obstruction, elective operation after stent insertion had similar oncologic outcomes compared with emergency operation. Preoperative stent insertion not only lowers the incidence of enterostomy but also makes primary anastomosis & laparoscopic surgery possible, thereby enhancing patients’ quality of life. Therefore, preoperative stent insertion is a useful method that may replace emergency surgery in treatment of malignant colorectal obstruction.
Notes
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This work was supported by the 2014 Inje University Busan Paik Hospital research grant.