후복막 신장주위에 발생한 국소성 유리질 혈관형 캐슬만병
Unicentric hyaline-vascular type Castleman’s disease in the pararenal retroperitoneum
Article information
Abstract
Castleman’s disease (CD) is a relatively rare and benign disorder by the proliferation of lymphoid tissue. Although they can occur either in the intraperitoneal or retroperitoneal spaces, CD in the pararenal retroperitoneum is especially rare, accounting for only 2% of all reported cases. Despite the recent advances in radiologic imaging modalities, CD is a rare entity that is difficult to make a correct preoperative diagnosis. But, I herein describe the case of a 48-year-old man, with hyaline-vascular type CD of the pararenal retroperitoneum preoperatively diagnosed by multidetector computed tomography and 18F-fluorodeoxyglucose positron emission tomography/computed tomography and successfully treated with curative resection of the mass. Postoperatively, the final diagnosis was confirmed by histologic examination.
INTRODUCTION
Castleman’s disease (CD) is a relatively uncommon disorder by the benign proliferation of lymphoid tissue, which was first described by Castleman et al. [1,2] in 1954. Although the lesions most commonly arise in the mediastinum (70%), they can occur in any region of the body where lymphoid tissue is normally present [3]. CD in the pararenal retroperitoneum is especially rare, accounting for only 2% of all reported cases [4]. Most of the retroperitoneal tumors are malignant lesions such as liposarcoma, leiomyosarcoma, and fibrohistiocytoma. But, clinically, benign tumors like CD are indistinguishable from the other malignant retroperitoneal tumors. So, preoperative clinical diagnosis of retroperitoneal CD is very important. Although many reports of CD are described preoperative diagnosis remains difficult, but I present a preoperatively diagnostic case with multidetector computed tomography (MDCT) and 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG-PET/CT) and successfully treated with curative resection of the mass. Especially, preoperative diagnosis of CD by MDCT is the second description since its first report by Zheng et al. [5] in 2008. Also, to my knowledge, this is the first reported case detected by 18F-FDG-PET/CT for hyaline-vascular type CD in pararenal retroperitoneum.
CASE REPORT
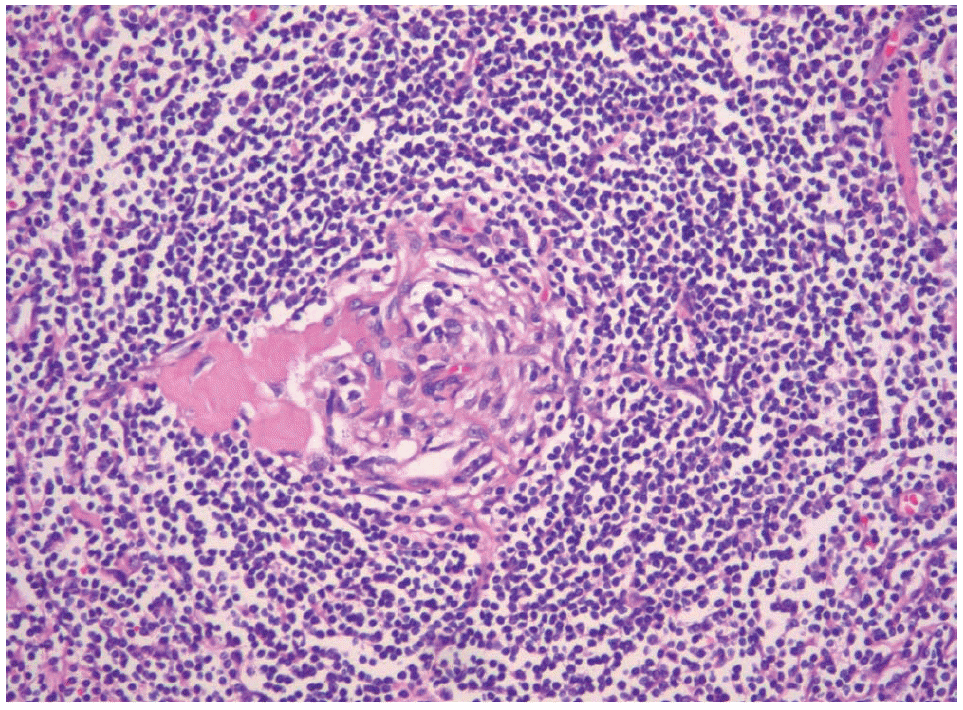
A 48-year-old man presenting with abdominal discomfort was referred to the our hospital. His medical and family history were unremarkable. He had no history of previous abdominal surgery. On admission, preoperative routine examination and tumor markers (carcinoembryonic antigen, carbohydrate antigen 19-9) had no signifiCase cant abnormalities. The precontrast abdominal CT imaging showed a well-demarcated and non-enhanced mass in the left pararenal area, measuring approximately 4.0×4.0 cm in diameter (Fig. 1A). At the postcontrast arterial imaging CT, the mass revealed a peripheral ‘rimlike’ enhancement at the margin of the tumor (Fig. 1B). In fasting state (serum glucose 87 mg/dL), the patient received 13.8 mCi of 18F-FDG intravenously. A whole-body 18F-FDG-PET/CT scan was performed at 1 hour after 18F-FDG injection. 18F-FDG-PET/CT scan showed a focus of radiotracer accumulation in the left pararenal mass. Standard uptake value (SUV) for the pararenal mass was 2.2 (Fig. 1C). From these radiographic findings, we diagnosed CD in the pararenal retroperitoneum and surgical excision was performed. At laparotomy, a solid retroperitoneal mass was identifed with adhesion to the adjacent tissue including the left kidney and renal artery. Owing to the severe adhesion, the patient had to be treated by an extended resection including left nephrectomy (Fig. 2). Operative time was 125 minutes and estimated blood loss was 100 mL. Grossly, the mass was an encapsulated round mass, measuring 3.6×3.0×2.5 cm in diameter. The cut surface of the mass was brownish yellow without areas of hemorrhage and necrosis (Fig. 2). Based on the histologic examination, the tumor was finally diagnosed as a hyaline-vascular type CD (Fig. 3). The patient had an uneventful postoperative course and no evidence of recurrence has been seen in the 48 months since his operation.

(A) The pre-contrast computed tomography (CT) imaging shows a homogeneous non-enhanced tumor in the left pararenal area (arrow). (B) The post-contrast arterial imaging CT shows a peripheral ‘rim-like’ enhancement arround the tumor (arrow). (C) 18F-fluorodeoxyglucose positron emission tomography/CT scan shows a focus of radiotracer accumulation (arrow).
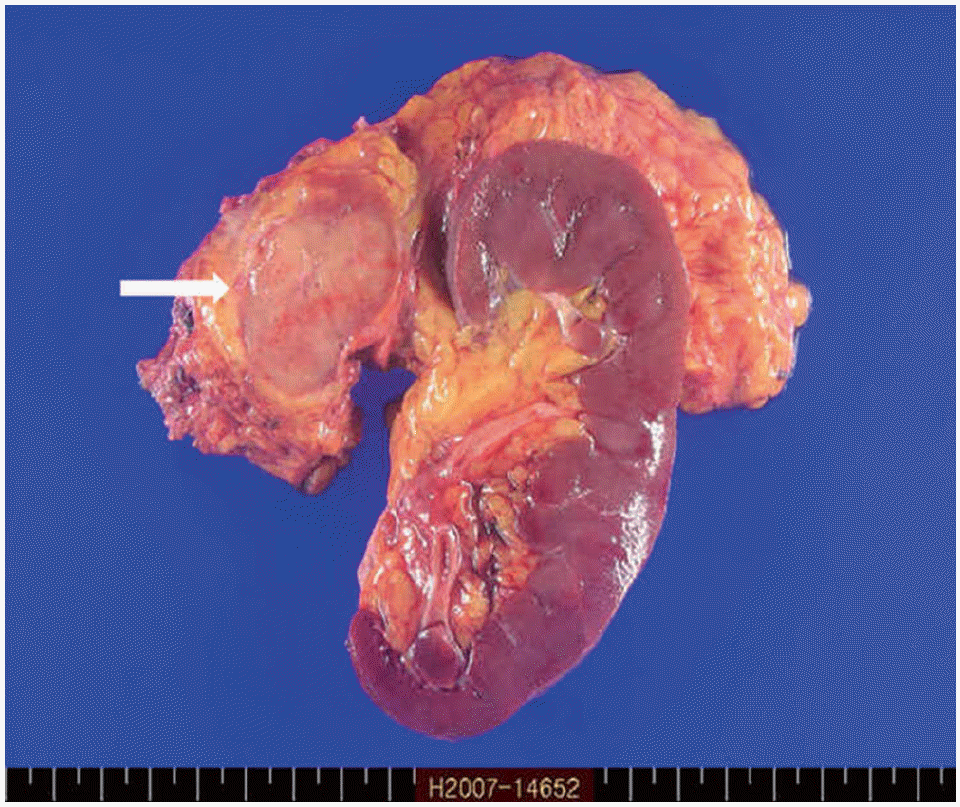
Grossly, a well defined and round mass (3.6×3.0×2.5 cm) is present in the surrounding perinephric adipose tissue. The cut surface of the mass is brownish yellow without areas of hemorrhage and necrosis (arrow).
DISCUSSION
CD is a relatively rare benign disorder characterized by the formation of lymph node masses, which was first described by Castleman et al. [1,2] in 1954. The etiology of this disease is uncertain, but may represent chronic inflammatory response [6]. There are two theories have been suggested. One theory submits that the disorder represents a reactive lymphoid hyperplasia, initiated by chronic antigenic stimulation associated with a viral trigger. The other one proposes that the masses are due to a developmental growth disturbance of the lymphoid tissue [5]. CD is usually classified into two clinical subtypes: unicentric and multicentric varieties [7]. Unicentric type is more common and manifests as a solitary mass that most often affects younger patients. Multicentric type has systemic symptoms and is relatively rare in children. CD can be histologically divided into two major types: the hyaline-vascular type, which accounts for more than 90% of the cases and the less common plasma-cell type [4]. Most cases with hyaline-vascular type are found by chance, whereas plasma-cell type may occasionally be the cause of fever from an unknown origin [7]. Approximately 70% of CD cases occur in the mediastinum and 20% in the cervical, axillary, inguinal, shoulder, and vulvar regions. Although they can occur either in the intraperitoneal or retroperitoneal spaces, CD of retroperitoneal origin is only 7% of all cases of this disorder. CD in the pararenal retroperitoneum is especially rare, accounting for only 2% of all reported cases [4]. The differential diagnosis for retroperitoneal tumors includes benign or malignant lesions. Most of the tumors are malignant lesions such as liposarcoma, leiomyosarcoma, and fibrohistiocytoma [6]. Benign tumors are relatively rare, comprising only about 20% of all primary retroperitoneal neoplasms and include leiomyoma, lipoma, xanthogranuloma, neurofibroma, lymphangioma, extraadrenal pheochromocytoma, retroperitoneal cyst, and urogenital ridge remnant [4]. Clinically, there are no distinctive features of retroperitoneal CD. Also, radiographically, CD is indistinguishable from other retroperitoneal tumors [8]. As reported in literature, unicentric hyaline-vascular type CD manifests on CT as a single, well-defined, and nonspecific homogeneous mass with marked or moderate enhancement. However, if the mass is larger than 5 cm in diameter, it will more likely be heterogeneous with central low-attenuation areas, which may account for fibrosis or necrosis [9,10]. Zheng et al. [5] reported new CT finding by MDCT. They discovered MDCT not only reveals a well-defined, homogeneous soft-tissue attenuation mass with marked enhancement, but also shows a peripheral ‘rim-like’ enhancement [5]. In this current case, the sign of peripheral ‘rim-like’ enhancement was shown at the arterial phase. Also, a limitation of CT cannot help distinguish between reactive hyperplasia and pathological enlargement. So, previous reports suggested that FDG-PET imaging modality can help differentiate benign CD from maligngnat tumor [11,12]. To my knowledge, previous studies of 18F-FDG-PET for CD have reported six cases of unicentric CD [7,11-15] and one case of multicentric CD [14]. Especially, current study of unicentric CD in pararenal retroperitoneum by 18F-FDG-PET/CT is the first case. Murphy et al. [15] reported that PET imaging of CD demonstrated FDG accumulation in a range lower than that seen for low-and intermediate-grade lymphomas, so suggested the use of FDG-PET as an imaging modality to differentiate benign CD from malignant lymphoma. In current case, FDG accumulation (SUV: 2.2) was low like previous report [15]. Toita et al. [7] reported that 18F-FDG-PET had a useful role in the detecting lesion localization in young case without any localized symptoms or signs or with very small lesions. In the unicentric forms of CD, complete surgical resection is the treatment of choice associated with very good prognosis after complete resection [5]. In this current case, the tumor had a severe adhesion to the adjacent tissue including the left kidney and renal artery. So, the patient had to be treated by an extended resection including left nephrectomy. In conclusion, we consider that peripheral “rim-like’ enhancement by MDCT can be very useful role in making a diagnosis preoperatively in CD, and 18F-FDG-PET/CT would be an imaging modality to differentiate benign CD from other malignant lesions.
Notes
No potential conflict of interest relevant to this article was reported.